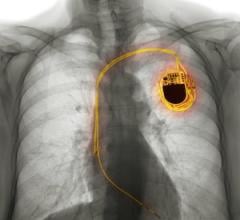
November 18, 2008 - According to a study published in the Nov. 18 issue of the journal Circulation, Medtronic's Lead Integrity Alert (LIA) significantly improves early identification of potential implantable cardioverter-defibrillator (ICD) lead fractures.
LIA offers added protection for ICD patients through more frequent audible alarms and is the first continuous ICD monitoring technology that triggers real-time device changes to reduce unnecessary shocks that could result from potential lead fractures, Medtronic said. Study results showed LIA provided at least a three-day advanced warning of unnecessary shocks in 76 percent of patients. In the analysis of about 16,000 patients, when the LIA alert was triggered, 72 percent of alerts were lead fractures and 81 percent of alerts were lead fractures or header connector problems requiring surgical intervention. The false-positive rate was one per 372 patient-years of monitoring.
The LIA received both FDA and CE Mark approval in September 2008. Pending further regulatory approvals, LIA will be included in all future Medtronic implantable defibrillators and applies to all Medtronic leads, including the Sprint Quattro Secure S lead (Model 6935), which was recently approved in Europe.
"The lead integrity algorithm is an important advance in diagnostics that monitor the integrity of defibrillation leads," said Charles D. Swerdlow, M.D., cardiac electrophysiologist, Cedars-Sinai Medical Center in Los Angeles and lead author on the study. "It is the first downloadable software developed to enhance the performance of normally-functioning ICDs and the first to trigger real-time changes in ICD parameters to reduce unnecessary shocks caused by lead fractures. Our study demonstrates that the lead integrity algorithm increased the warning time from diagnosis of lead fracture until the patient receives unnecessary ICD shocks caused by the fracture. This algorithm can prevent unnecessary shocks if the patient and physician respond rapidly once an alert is triggered."
LIA was developed as a software upgrade for nearly all (98 percent) Medtronic ICDs and cardiac resynchronization therapy-defibrillators (CRT-Ds) implanted in the U.S., and 93 percent of these implanted worldwide.
The ability of LIA to increase the time between a lead fracture and potential delivery of an unnecessary shock is based on several factors, Medtronic said. These include improved sensing capabilities to help detect possible lead fractures earlier and increased number of intervals required to detect VF (following detection of a lead fracture) to avoid treating lead fractures as VF. The system also has an increased number of times the audible alert sounds each day so that patients are more likely to hear the alert. Upon hearing the alert, patients should contact their physician; and expanded electrogram storage to help clinicians diagnose the cause of the alert.
For more information: www.medtronic.com, www.CRDMPPR.medtronic.com

 April 24, 2026
April 24, 2026 









