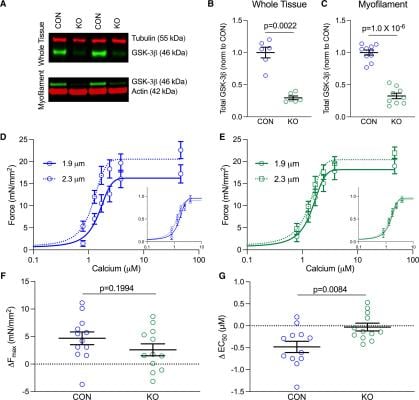
Figure 1. Genetic ablation of GSK-3β (glycogen synthase kinase 3β) results in loss of length-dependent activation. A, Example western blots of GSK-3β in left ventricular (LV) whole tissue lysis ( top) and myofilament-enriched ( bottom) samples from GSK-3β knockout (KO) and littermate control (CON) mice. B, Quantification of GSK-3β in whole tissue (P=0.0022, n=6/group) and ( C) myofilament-enriched (P=1.0×10−6, n values: CON=10, KO=9) samples. GSK-3β was normalized to either tubulin (whole tissue) or actin (myofilament) and further normalized to CON, shown as mean±SEM. P values were calculated by Mann-Whitney t test for the whole tissue comparison. D, Mean force as a function of calcium concentration and fitted curves for skinned myocytes from CON and ( E) KO mice in which curves were measured at a sarcomere length (SL) of 1.9 µm and then 2.3 µm. Normalized curves are shown in the bottom right of each graph to emphasize shifts in calcium sensitivity. F, Delta Fmax and ( G) EC50 for CON and GSK-3β KO mice between SL 1.9 and 2.3 µm (P=0.0084 by unpaired t test, n=12 myocytes from 4 CON and 3 KO mice).
March 22, 2022 – In an article published in Circulation Research, a leading cardiovascular research journal, a team of researchers at Loyola University Chicago Stritch School of Medicine identified a novel mechanism by which the heart functions, potentially leading to the discovery of a therapeutic to treat adults who experience heart failure. According to the U.S. Centers for Disease Control, 6.2 million Americans experienced heart failure in 2018.
“For years, researchers have been piecing together factors that contribute to contraction of the heart, which stems from a complex structure in each heart cell called the sarcomere,” said Marisa Stachowski, a PhD candidate at Stritch and lead author of the study. “Through our research, we discovered that the enzyme glycogen synthase kinase 3 beta (GSK-3beta) binds to the sarcomere and helps regulate how the sarcomere, the molecular motor that drives contraction, functions.”
Heart failure occurs when the heart cannot pump sufficient blood to the organs throughout the body, and has many causes, including coronary artery disease, diabetes, high blood pressure, obesity, and genetics. “Heart failure is a complex syndrome,” explains Jonathan Kirk, associate professor at Loyola and senior author of the study, “but we know the sarcomere plays a central role in almost all forms of the disease. For this reason, my lab works to identify potential molecular targets to modify and improve sarcomere function and improve heart health.”
Normally, the molecular motors in the sarcomere respond to how much blood fills the heart: the more blood, the more the cells are stretched, and the stronger the contraction to pump this blood throughout the body. This behavior, known as the Frank-Starling mechanism, has been known for over a century, but how it works at the molecular level is still mostly a mystery. The research conducted by Stachowski and the team showed that heart failure patients had reduced levels of GSK-3beta bound to the sarcomere and were missing a normal Frank Starling mechanism. Importantly, restoring sarcomere GSK-3beta rescued the Frank Starling mechanism.
“The challenge,” explained Stachowski, “is that the enzyme, GSK-3beta, has a lot of jobs in each heart cell, so how do we direct it to the sarcomere? Through our research, we found a switch called phosphorylation, that causes GSK-3beta to go to the sarcomere and modify the Frank-Starling mechanism.” By creating mutant versions of the protein, Stachowski and colleagues showed that they could specifically direct GSK-3beta to go to the sarcomere. This important discovery provides a tool for restoring the heart’s ability to contract and relax, and a potential therapeutic target to treat heart failure.
The study is titled “GSK-3β Localizes to the Cardiac Z-Disc to Maintain Length Dependent Activation.” In addition to Stachowski and Kirk, other co-authors include: Dr. Maria Papadaki, Dr. Thomas G. Martin, Nitha Aima Muntu, and Edith Perez from Loyola University Chicago; Dr. Weikang Ma, Henry Gong, and Dr. Thomas Irving from Illinois Institute of Technology; Stephanie Shao, Shi Shen, and Dr. Stuart G. Campbell from Yale University; Dr. Mohit Kumar, and Dr. Sakthivel Sadayappan from University of Cincinnati; Dr. Jody L Martin from UC Davis; Dr. Christine S Moravec from Cleveland Clinic. The study was funded by grants from the National Institutes of Health, National Science Foundation, and the American Heart Association.
For more information: https://ssom.luc.edu/


 May 13, 2026
May 13, 2026 









