
The Philips AAA Model integrates software and Philips 3-D ultrasound technologies into a single solution to automatically segment and quantify the size of the aneurysm sac for surveillance of known native and post-EVAR treated AAAs.
January 27, 2021 — Philips Healthcare has introduced the Philips Abdominal Aortic Aneurysm (AAA) Model, providing physicians a more patient-friendly solution compared to the current standard of care for managing AAA patients. Based on 3-D ultrasound, Philips AAA Model delivers clinicians accurate diagnostic information without exposing patients to high doses of radiation and nephrotoxic contrast agents.
An abdominal aortic aneurysm (AAA) is an aneurysm that forms in the lower part of the aorta. Typically, AAAs are identified incidentally during abdominal imaging exams but, in some cases, remain undetected until rupture. A ruptured AAA has an 80% mortality rate,[1] emphasizing the importance of routine surveillance.
The Philips AAA Model integrates innovative software and leading Philips 3-D ultrasound technologies into a single solution to help increase diagnostic confidence and an improved patient experience. The software automatically segments and quantifies the size of the aneurysm sac for surveillance of known native (untreated), and post-EVAR (endovascular aortic repair — treated) AAAs.
Current Standard of Care to Image AAAs is CT and 2-D Ultrasound
The current standard of care for AAAs includes 2-D ultrasound and computed tomography angiography (CTA). Each of these modalities has its drawbacks, including inter-operator variability with 2-D ultrasound and patient exposure to high levels of radiation and nephrotoxic contrast agents with CTA.
A recent clinical study showed that 3-D ultrasound examination for native AAA surveillance has excellent inter-operator reproducibility, superior to that of 2-D ultrasound, supporting the broader use of 3D ultrasound in standard AAA surveillance programs.[2] 3-D ultrasound has been shown to estimate the diameter and volume of an AAA with acceptable reproducibility and an improved agreement (over 2-D ultrasound) with CT [3]. Furthermore, 3D ultrasound has also been proven to correlate significantly better to 3-D CT than 2-D ultrasound for assessing the maximum diameter of the residual sac post-EVAR, with clinically acceptable reproducibility.[4]
Philips Abdominal Aortic Aneurysm (AAA) Model seamlessly integrates leading Philips technologies including Philips Premium Ultrasound System (Epiq Elite), Philips Array Transducer (X6-1 xMatrix) and innovative software into a single solution, to help improve the experience and outcomes for clinicians and patients alike.
“Regular surveillance of abdominal aortic aneurysm patients is essential, but today’s standard of care has downsides,” said Bich Le, senior vice president, general manager ultrasound at Philips. “Philips Abdominal Aortic Aneurysm (AAA) Model seamlessly integrates leading Philips technologies including Philips Premium Ultrasound System, Philips Array Transducer and innovative software into a single solution.”
Launch of Philips Abdominal Aortic Aneurysm (AAA) Model at LINC 2021
Philips will debut the AAA Model at the LINC (Leipzig Interventional Course) Summit, Europe’s premiere interventional course for vascular specialists, taking place virtually, Jan. 25-29, 2021. Visit Philips at LINC for more information on Philips AAA model, including live demos, and follow @PhilipsLiveFrom for updates throughout the virtual event. LINC attendees are also invited to attend a Symposium on Aortic Aneurysms, Friday, Jan. 29 at 12:30 PM CET, where clinicians will discuss the highest quality of care in complex EVAR procedure with advanced imaging techniques, from planning to follow-up. Participating in the Symposium is Prof. Henrik Sillesen who will address improving AAA measurement accuracy using innovative Ultrasound 3D volume moderation and xMatrix.
The Philips Abdominal Aortic Aneurysm (AAA) Model is CE marked for sale in Europe and is FDA cleared for sale in the U.S.
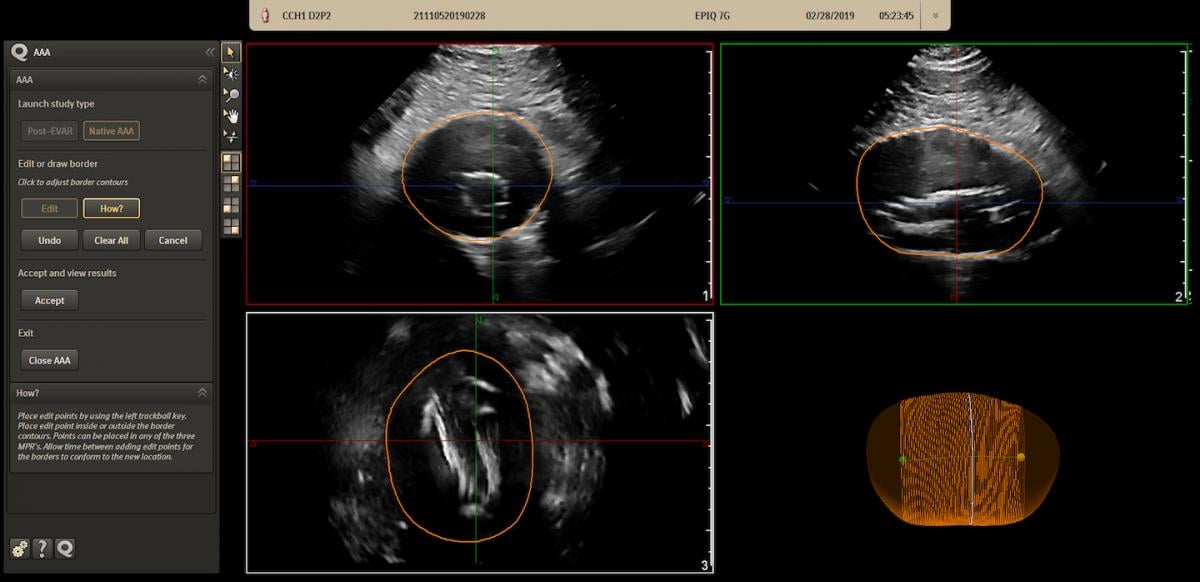
Philips Epiq AAA ultrasound assessment software can help diagnose and monitor aortic aneurysms.
For more information: www.philips.com.lb/en/healthcare/resources/landing/ultrasound/abdominal-aortic-aneurysm-model
References:


 May 12, 2026
May 12, 2026 









