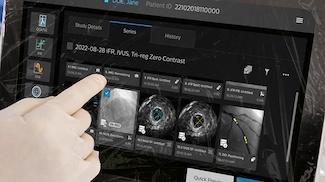
Philips IntraSight Plus (Photo: Philips)
March 25, 2026 — Royal Philips has launched its intuitive interventional guidance platform, IntraSight Plus, which is cleared for clinical use in the U.S. and Europe. With CE marking and FDA 510(k) clearance, the redesigned platform combines the most comprehensive diagnostic and treatment planning tools to deliver efficiency, simplicity and precision in a single intuitive system1. By streamlining access to key interventional tools, IntraSight Plus enables clinicians to diagnosis, virtually plan treatment, guide device placement and verify results on a single screen — supporting confident decision-making and improved patient outcomes.
Interventional cardiologists have traditionally relied on angiography alone for percutaneous coronary interventions (PCI), a common minimally invasive procedure. While guidelines support the use of intravascular ultrasound and physiology for PCI, systems and inputs are often separate, making adoption seem more challenging and less efficient in busy cath labs. IntraSight Plus aims to help with these challenges, bringing everything to the table through a single intuitive system that can help deliver 47% system operation time savings and enable complete control at the bedside2.
“As more advanced technology is integrated into the cath lab, the need for connectivity between the imaging tools is more important than ever before,” said Stacy Beske, Business Leader Image Guided Therapy Devices at Philips. “IntraSight Plus was built using feedback from our customers and represents a fundamental shift in interventional cardiology – a future driven by integrated intelligence that lets physicians focus on the decision-making that only they can do.”
Simplifying Complex Interventions
On March 19, 2026, Prof. Dr. Jan-Malte Sinning performed the first patient case using IntraSight Plus at Cellitinnen-Krankenhaus St. Vinzenz in Köln, Germany. The hospital recently renewed its catheterization lab infrastructure with Philips’ Azurion interventional suites. The addition of IntraSight Plus as part of this ecosystem allows for enhanced physician workflow.
“Modern PCI is becoming increasingly complex,” said Prof. Dr. Sinning. “Intravascular imaging combined with physiology provides the level of detail we need for contemporary PCI. What stands out with IntraSight Plus is that the platform is clearly designed around how we actually work in the cath lab — combining physiology, imaging and decision-making support in a way that fits naturally into the clinical workflow.”
The patient treated in this first case presented with an acute ST-elevation myocardial infarction (STEMI) of the left anterior descending (LAD) artery two days ago and underlying complex three-vessel disease. Using the Philips Azurion interventional platform in combination with the new IntraSight Plus system, Prof. Dr. Sinning performed a complete revascularization prior to discharge.
The integrated tri-registration of angiography, physiological assessment, and intravascular imaging enabled a streamlined and intuitive workflow, allowing treatment to be focused on hemodynamically relevant lesions and ensuring optimal stent deployment to improve patient outcomes.
Delivering Precision Through Integration
A fully redesigned system that merges Philips IntraSight and SyncVision technologies, IntraSight Plus brings Class IA IVUS and instantaneous wave-free ratio/fractional flow reserve (iFR/FFR) physiology, co-registration, tri-registration and real-time device visualization together in one system.
It delivers:
-
Greater Precision: Class IA IVUS and iFR tri-registered onto one screen so clinicians can see the plaque morphology and its ischemic impact at every point along the vessel. Physicians can plan their PCI, estimate results, size and place stents and perform precise PCIs for better patient care.
-
Efficiency: Enter patient data once. Streamlined communication between IntraSight Plus and Azurion DataHandler is designed for customers to spend less time operating multiple technologies3.
-
Customer Control: The intuitive platform brings procedural guidance and unified control to the bedside, providing complete bedside procedural control without leaving the sterile field.
Philips’ portfolio of image-guided therapy solutions uniquely integrates best in class imaging systems and software, with specialized diagnostic and therapeutic devices to support exceptional treatment for even the most complex procedures. Built using a common Philips architecture, IntraSight Plus also connects with the company’s Azurion platform and PACS system for tighter integration within a Philips lab environment.
Availability
IntraSight Plus is FDA 510(k) cleared in the USA and CE marked in Europe. Commercial availability is subject to market release and applicable regulatory requirements.


 April 20, 2026
April 20, 2026 









