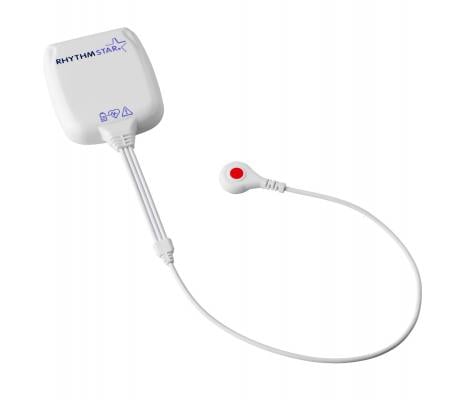
Remote cardiac monitoring vendor RhythMedix released its next-generation RhythmStar wearable device with built-in 4G cellular connectivity. The cardiac telemetry monitor can be worn for extended remote monitoring without the need for an additional phone or communication device.
February 23, 2021 — Remote cardiac monitoring vendor RhythMedix released its next-generation RhythmStar wearable device with built-in 4G cellular connectivity. The novel cardiac telemetry monitor is discreetly worn for extended remote monitoring without the need for an additional phone or communication device.
The technology enables rapid ECG analysis and reporting of cardiac abnormalities by RhythMedix's team of U.S.-based certified technicians, who alert physicians if an arrhythmia of concern is detected. RhythmStar is now available with HeartTime™ on-demand telemetry mode, an innovative platform that allows physicians to evaluate cardiac activity and expedite care via a telemedicine appointment, limiting the need for an in-person office visit.
Heart arrhythmias are a major health concern that doubles the risk of death in an estimated 33.5 million people worldwide.[1,2] While remote cardiac monitoring has improved arrhythmia detection, patient compliance and timely access to heart activity currently limit accurate and timely diagnosis.
"The novel wearable RhythmStar monitoring device offers increased patient compliance and allows for accurate assessment of cardiac rhythm in real time," said Steven DaTorre, M.D., FACC, a clinical cardiologist at Cooper University Health Care in New Jersey. "It also allows me to remain connected to my patients throughout their monitoring period, which may expedite a life-saving diagnosis."
The sleek, one-piece device, with built-in 4G cellular technology, is the first wearable multi-modality cardiac monitor (Holter, event and MCT) that connects patients and clinicians for improved patient care. RhythmStar integrates seamlessly into clinical practice, helping physicians make informed treatment decisions that allow patients to enjoy life, without missing a beat.
"We are excited to offer physicians and their patients our unique monitoring solution that improves patient compliance, helps accelerate diagnosis, and allows physicians to stay virtually connected to their patients with on-demand access to clinical data", stated Brian Pike, president, chief operations officer and co-founder of RhythMedix. "We look forward to bringing this next-generation RhythmStar technology to physicians across the U.S. in 2021."
For more information: www.rhythmedix.com
References:


 March 04, 2026
March 04, 2026 









