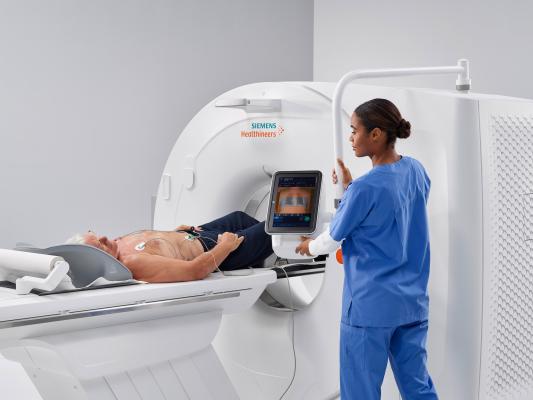
September 17, 2018 — The Ohio State University Wexner Medical Center in Columbus recently became the first healthcare facility in the United States to install the new Somatom go.Top computed tomography (CT) system from Siemens Healthineers. The 128-slice scanner expands the Somatom go. CT platform’s concept of patient-centric mobile workflow – which is controlled via tablet and remote – into advanced clinical fields and applications, including cardiology, CT-guided intervention and dual-energy spectral imaging.
In addition to a 0.33-second rotation time, Stellar detector technology and a robust 75 kW generator, the Somatom go.Top has a large detector with an acquisition speed of up to 175 mm in one second and an automated workflow. The system can deliver new levels of radiation dose personalization due to its high power and low kV scanning mode, along with automated tools that take the guesswork out of low-dose CT imaging. And with the system’s mobile, tablet-based workflow and the capability to off-load manual reconstruction tasks, technologists can spend more time focused on the patient.
Explaining his facility’s decision to select the Somatom go.Top, Richard D. White, M.D., chairman of the Department of Radiology at The Ohio State University Wexner Medical Center, said, “We were looking for an application of robust, tablet-based capabilities facilitating direct patient engagement and partnership while leveraging automated workflows to significantly reduce exam variability and error risk, as well as increase patient-specific image quality.”
The Ohio State Wexner Medical Center is also using the system’s Twin Beam Dual Energy capabilities to perform dose-neutral dual energy scans that support oncological workups for patients receiving cancer care. Finally, the Ohio State team is using a new feature of Somatom go. scanners – Guide&GO, the first tablet-based solution for CT-guided interventional work – to enable touch-screen control in the intervention suite.
For more information: www.usa.healthcare.siemens.com


 May 12, 2026
May 12, 2026 









