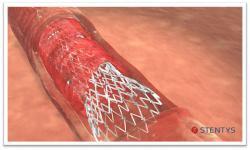
June 18, 2015 - Stentys announced in April that it received CE Marking for its new Self-Apposing stent system ahead of plan. The designation will allow the company to market the new products in Europe and in all the other countries where this certification is recognized. The Xposition S stent officially launched at the EuroPCR conference on May 19, 2015.
Xposition S is Stentys' next-generation sirolimus-eluting self-apposing stent. It is delivered by a system that closely replicates conventional stent implantation techniques, making the implantation of a Stentys stent easier and requiring practically no training.
The delivery system was designed based on the Cappella Peel Away technology that the company acquired less than a year ago, where a small balloon splits open the sheath containing the stent to release it precisely at the intended location.
For more information: www.stentys.com


 May 13, 2026
May 13, 2026 









