June 8, 2022 — Us2.ai, a Singapore-based medtech firm backed by, IHH Healthcare, Heal Partners, Sequoia India, EDBI, Pappas Capital and Partech Ventures has received CE Mark clearance for Us2.v1, a completely automated decision support tool for echocardiography. This follows US FDA clearance in 2021 and Health Sciences Authority Singapore clearance in 2022.
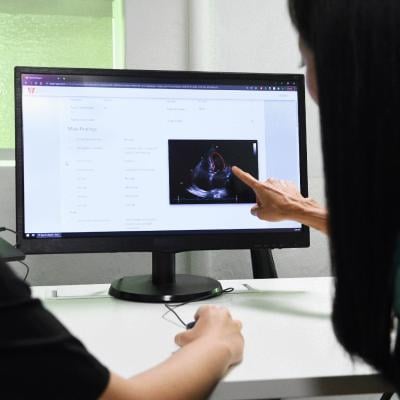
Us2.ai is pleased to announce that Us2.v1 is now available for clinical use in UK and across Europe, US, Canada, Australia, New Zealand and Singapore. Us2.v1 is a patented, automated clinical workflow solution that recognizes and analyzes 2-dimensional and Doppler echo images for comprehensive cardiac measurements needed for the diagnosis, prediction and prognosis of heart disease and pulmonary hypertension.
"These approvals for clinical use across Europe, in the UK and in our home country of Singapore will help accelerate the roll out of Us2.v1 as an important support system for cardiologists,” said James Hare, CEO and co-founder of Us2.ai.
“Heart disease is the leading cause of death globally. Us2.v1 creates a complete and fully automated patient report with editable annotations, conclusions and comparisons to international reference guidelines to ensure cardiologists are armed with all the facts to detect heart issues early and with ease,” added James.
Us2.ai has also announced the receipt of a Notice of Allowance, from the US Patent and Trademark Office, with the title: Clinical Workflow to Diagnose Heart Disease Based on Cardiac Biomarker Measurements and AI Recognition of 2D and Doppler Modality Echocardiogram Images.
"This is important because multi-modality AI analytics, combining imaging and circulating biomarkers, is key for precision approaches in cardiology,” said Dr Carolyn Lam, Senior Consultant Cardiologist at National Heart Centre Singapore, Professor at Duke-National University of Singapore and co-founder of Us2.ai.
Us2.v1 automated measurements include 2-dimensional (cardiac volumes, all 4 chambers of the heart), M-mode (e.g. tricuspid annular plane systolic excursion), spectral Doppler (blood flow across all valves, both PW and CW measurements) and tissue Doppler; thus covering the vast majority of standard measurements for adult transthoracic echocardiography recommended by the American Society of Echocardiography, European Association of Cardiovascular Imaging, and British Society of Echocardiography. Fully automated Us2.v1 measurements were shown to be completely interchangeable with expert human measurements. Furthermore, Us2.v1 measurements were completely reproducible for a given patient study, with image processing/analysis algorithm computation time of approximately two minutes per study.2
For more information: https://us2.ai/
References:
1 Measurements validated at the Brigham & Women’s Hospital:
Left ventricle: DecT, MV-A, MV-Adur, MV-E, e’ lateral, e’ septal, a’ lateral, a’ septal, s’ lateral, s’ septal, LVEDV MOD biplane, LVEF MOD biplane, LVESV MOD biplane, LVSV MOD biplane, IVSd, LVIDd, LVIDs, LVPWd, E/e’ mean
Left atrium: LAESV MOD biplane
Right ventricle: RVIDd
Right atrium: RAa
Tricuspid valve: Tr Vmax
2 Total time for interpretation of a study can depend on other factors such as the interpreting physician and preparation for uploading DICOMs for analysis.


 April 28, 2026
April 28, 2026