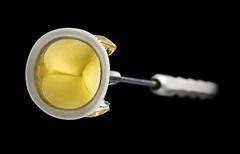
April 22, 2011 – The U.S. Food and Drug Administration (FDA) has cleared the Trifecta valve, by St. Jude Medical. The valve, a clinically-proven replacement for diseased, damaged or malfunctioning aortic heart valves, mimics the flow of a natural, healthy heart. The valve was designed for excellent hemodynamic performance (the optimization of blood flow through the valve) and long-term durability. To ensure the structural integrity of the valve, it is constructed using a polyester and tissue-covered titanium stent, or base. The valve features leaflets manufactured from pericardial tissue attached to the exterior of the valve stent. This design allows the leaflets to open more fully and efficiently, mimicking the performance of a healthy aortic heart valve and limiting tissue abrasion through tissue-to-tissue (stent-to-leaflet) contact. Also contributing to the valve’s durability, it offers the Linx AC Technology, an anticalcification treatment designed to reduce tissue mineralization (hardening), one of the primary causes of valve deterioration. It also offers several features that improve the ease of implantation, including the valve’s unique holder, designed to improve visibility and cuff access for suturing, and customized sizes. “Physicians around the world have already voiced tremendous excitement and satisfaction with the Trifecta valve,” said Frank J. Callaghan, president of the St. Jude Medical cardiovascular division. “Trifecta has demonstrated outstanding hemodynamic performance and has improved the lives of patients with aortic valve disease.” The valve was approved by regulatory authorities in Europe and Canada in 2010. For more information: www.sjm.com


 March 25, 2026
March 25, 2026