October 6, 2023 — Biosense Webster, Inc., a global leader in cardiac arrhythmia treatment and part of Johnson & Johnson MedTech,1 announced new data from a Biosense Webster funded study which sought to compare the risk of heart failure incidence among atrial fibrillation (AFib) patients treated with catheter ablation versus antiarrhythmic drugs (AAD). The study, titled “Heart failure risk in patients with atrial fibrillation treated with catheter ablation versus antiarrhythmic drugs,” was published in Heart Rhythm O2, and evaluated non-specific catheter claims information and found that catheter ablation was associated with significantly lower risk of incident heart failure compared to treatment with AAD only.i
“The results from this study underscore the positive, life-changing impact catheter ablation can have for people with AFib,” said Jasmina Brooks, President, Biosense Webster, Inc. “At Biosense Webster, we are proud of our legacy of helping electrophysiologists elevate the standard of care and are continuing to generate scientific evidence to advance new solutions and approaches to help those with AFib live the lives they want.”
Catheter ablation and AAD are two types of treatments that are widely used to treat patients with AFib. Several clinical studies have suggested that catheter ablation is a safe and superior alternative to AAD when performed appropriately, yet currently less than 5% of the population eligible for catheter ablation receives the procedure.1
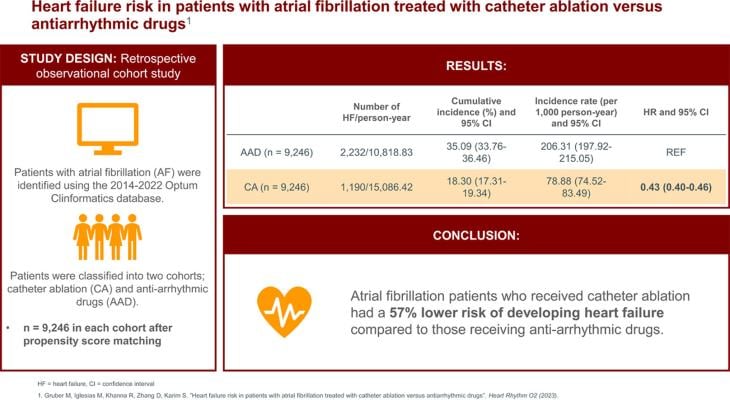
Using the 2014-2022 Optum Clinformatics database, the study identified adult patients with AFib who had used AAD previously. They were then classified into two cohorts: patients who were treated with catheter ablation versus patients who received an AAD. The two groups were then matched on socio-demographic and clinical covariates using propensity score matching technique. There were 9,246 patients in each of the two matched cohorts for AAD and catheter ablation, respectively.
The findings were statistically significant in showing patients receiving catheter ablation had a 57% lower risk of incident heart failure compared to those treated with AAD (hazard ratio [HR]=0.43; 95% CI=0.40-0.46).1 The lower risk of heart failure associated with catheter ablation treatment was consistent across all race/ethnicity and sex categories evaluated.
“In this examination of a large real-world dataset, we found that there was a significantly lower risk of heart failure for those patients undergoing cardiac ablation treatment versus AAD as second line treatment,” said Saima Karim, DO, FACC, FHRS, Assistant Professor, Case Western Reserve University School of Medicine and Primary Research Partner in the Heart Failure Study.2 “For the many millions of patients impacted by AFib and the clinicians who treat them, these results provide compelling decision-making evidence for effective AFib treatment options when considering the incidence of new onset heart failure.”
AFib is the most common type of cardiac arrhythmia and affects more than 6 million people in the United States and over 37.5 million people worldwide.ii,iii Approximately 1 in 4 adults over the age of 40 are at risk for developing AFib.iv Individuals with AFib are at a higher risk of heart failure, a health condition that makes the body unable to pump blood as efficiently as it should.v
For more information: www.biosensewebster.com


 April 24, 2026
April 24, 2026