December 26, 2023 — Pulse Biosciences, Inc., a company primarily focused on leveraging its novel and proprietary CellFX Nanosecond Pulsed Field Ablation (nsPFA) technology for the treatment of atrial fibrillation, announced the completion of the first five procedures in its first-in-human feasibility study with its novel CellFX nsPFA cardiac catheter. All patients were successfully discharged by treating physicians. Patients will continue to be monitored and evaluated over the coming months to assess safety and effectiveness with the primary safety endpoint at 30 days.
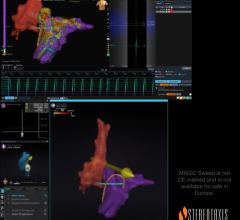
Dr. Vivek Reddy, Director of Cardiac Arrhythmia Services at Mount Sinai Hospital, NY, and Dr. Petr Neuzil, Chief of Cardiology at Na Homolce Hospital, Prague, and colleagues used the Company’s CellFX nsPFA 360 cardiac catheter integrated with 3D mapping and navigation technologies (iMap System, CardioNXT) to successfully treat five patients with atrial fibrillation (AF) at Na Homolce Hospital, a renowned research institution hospital in Prague, Czech Republic.
“We have been collaborating with Pulse Biosciences to bring their novel nsPFA technology to the clinical realm, and are excited to report that our experience with these first five patients has validated our belief that this may represent the next generation of PFA technology for the treatment of AF,” said Dr. Reddy. “The results were consistent with our preclinical experience. Importantly, the speed and ease with which we were able to isolate the pulmonary veins with the nsPFA 360 catheter was impressive and all patients tolerated the procedure well. Now we look forward to completing enrollment in this study to fully assess the safety and durability of nsPFA treatment.”
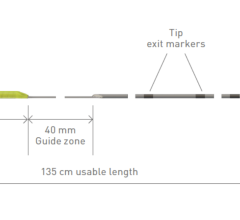
Pulse Biosciences’ CellFX nsPFA 360 cardiac catheter, which is still in the investigational stage, is uniquely designed to produce a nonthermal ablation, initially targeted for pulmonary vein isolation, using the Company’s proprietary CellFX nsPFA energy in the treatment of atrial fibrillation. The catheter is designed to deliver a fast, transmural and fully circumferential ablation in a single energy delivery. The CellFX nsPFA cardiac catheter is integrated with 3D mapping and navigation to deliver a comprehensive visualization and precise ablation delivery solution.
“We’re honored to work with Dr. Reddy and his team and Prof. Petr Neuzil and his team at Na Homolce to bring our next generation nsPFA technology to the clinic. The unique value of nsPFA to deliver fast, precise, transmural and contiguous ablations in thick cardiac tissue is extremely exciting for the treatment of AF and we could not have found better partners,” said Dr. Gan Dunnington, Chief Medical Officer, Cardiac Surgery of Pulse Biosciences. “This initial clinical experience delivered as expected. We look forward to continuing our catheter clinical program and to starting our cardiac surgery clinical program with our surgical nsPFA clamp in 2024.”
Kevin Danahy, President and CEO of Pulse Biosciences, having attended all patient cases, remarked on the recent milestone, “As we embark on this new era of advancement in medical device technology, our mission is clear: to revolutionize healthcare with CellFX nsPFA, with the intention of significantly improving clinical outcomes for both patients and physicians. Through relentless innovation, unwavering dedication, and commitment to pushing the boundaries of what is possible in bioelectric energy, we are reshaping the future of the treatment for atrial fibrillation with our cardiac catheter in electrophysiology and with our cardiac clamp in cardiothoracic surgery. CellFX nsPFA will empower doctors, inspire patients to seek life-altering treatment, and create a world where CellFX nsPFA technology can become a catalyst for healing and hope.”
The Company expects to enroll up to a total of 30 patients in the current feasibility study. Treated patients will be evaluated at regular intervals to further assess the safety and effectiveness of the treatments. The Company expects to provide additional updates on the first-in-human procedures with the CellFX system in the upcoming months. Pulse Biosciences will inform stakeholders promptly if results differ materially from the stated expectations addressing safety and efficacy. The observations to date have been positive but the broad set of risks associated with cardiac surgery remain. The Company anticipates initiating the regulatory process with the U.S. FDA and appropriate regulatory authorities worldwide including Europe in the coming quarters and expects additional studies will be required.
For more information: www.pulsebiosciences.com


 May 12, 2026
May 12, 2026