Photo: Solo Pace Inc.
March 31, 2026 — Solo Pace Inc. has announced the commercial release of the SoloPace Fusion Temporary Pacing System. The launch follows US Food and Drug Administration (FDA) 510(k) clearance for the system.
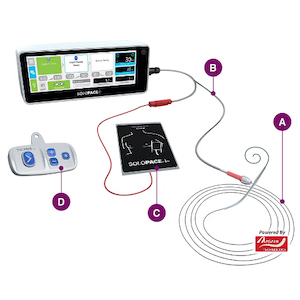
SoloPace Fusion is a single use, sterile kit designed to streamline temporary intracardiac pacing for transcatheter aortic valve replacement (TAVR) and other THV procedures. By combining guide wire functionality, left-ventricular pacing and patient grounding in one device, SoloPace Fusion delivers a standardized, predictable pacing method intended to simplify setup and reduce variability, without compromising smooth delivery and stability during valve deployment. As TAVR volumes increase globally, the need for efficient and effective technologies to complement valve therapies has become critical.
For interventional cardiologists using conventional right-ventricular (RV) pacing leads, SoloPace Fusion can eliminate the need for RV lead placement and the additional venous access it requires, potentially reducing procedure time and complication risk. For those using improvised LV pacing, SoloPace Fusion replaces ad-hoc clip assemblies, subcutaneous needles, and exposed wire connections – known sources of inconsistent pace capture and workflow variability.
"In our initial experience with SoloPace Fusion we've been able to shift from conventional RV pacing during many of our BE and SE TAVR procedures. RV pacing introduces risks for our patients, such as perforations and venous access issues, not to mention the time it adds to procedures. Never before have we had the combination of reliable LV pacing combined with smooth tracking and stability for valve deployment in one guidewire," said Michigan Health's Director of the Cardiac Catheterization Laboratory, Dr. Stanley J. Chetcudi, MD.
SoloPace Fusion is designed for use with the existing SoloPace Control external pulse generator (EPG) which is engineered to increase workflow efficiency during TAVR procedures while decreasing the risk of communication errors that can result in pacing complications. The system standardizes temporary pacing with operator-specific protocols, automated pace capture checks, sterile remote control operation and simple visual indicators.
"Clearance of our SoloPace Fusion System is a major milestone for our company and reflects our continued commitment to developing effective and efficient solutions for the growing number of Transcatheter Aortic Valve Replacement (TAVR) procedures done each year in the United States," said Solo Pace CEO, David Daniels, MD. "Our unique LV temporary pacing solution eliminates the need for improvised workarounds without compromising necessary guidewire performance. It will not only drive greater cath lab efficiency, but has the potential to deliver safer TAVR procedures for patients."
Additional information is available at www.solopace.health.


 April 20, 2026
April 20, 2026 









