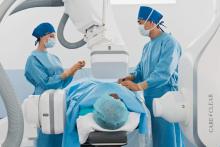
Physicians at the Heart Hospital of Austin, enrolled and successfully treated the first patient in the Roadster study, a global, multicenter clinical trial evaluating the safety and efficacy of the Silk Road System for the treatment of carotid artery disease. Mazin I. Foteh, M.D., a vascular surgeon and principal investigator for the trial, performed the first procedure.
© Copyright Wainscot Media. All Rights Reserved.
Subscribe Now