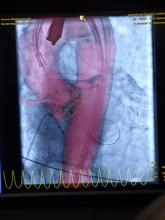
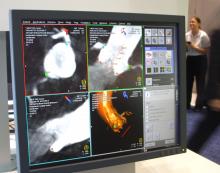
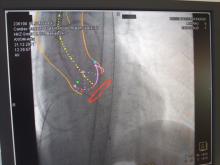
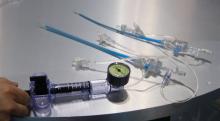
March 29, 2012 — Non-cardiac co-morbidities such as chronic obstructive pulmonary disease, chronic kidney disease and frailty are the main predictors of late mortality after transcatheter aortic valve implantation (TAVI), suggesting that patients with these conditions merit closer evaluation and follow-up, according to research presented at the American College of Cardiology’s (ACC) 61st Annual Scientific Session.
© Copyright Wainscot Media. All Rights Reserved.
Subscribe Now