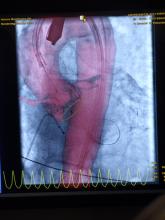
March 14, 2017 — Transcatheter aortic valve replacement (TAVR) for aortic stenosis continues to evolve with the adoption of methods intended to maximize efficacy, safety and patient recovery. Researchers at the Valve Science Center at Minneapolis Heart Institute Foundation recently conducted quality improvement review to examine the impact of the use of minimal anesthesia with conscious sedation (CS) in patients with severe, symptomatic aortic stenosis (AS) undergoing TAVR.
© Copyright Wainscot Media. All Rights Reserved.
Subscribe Now