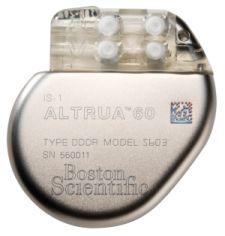
Boston Scientific's ALTRUA family of pacemakers is the first Boston Scientific-branded pacemaker to treat bradycardia.
The ALTRUA pacemaker provides physicians with a number of diagnostic and therapeutic capabilities designed to enable them to tailor the therapy to specific patient requirements. Some of the main features include multiple atrial ventricular (AV) delay programming options; minute ventilation (MV) blended sensor; ventricular rate regulation (VRR); and automatic capture for ventricular pulse management. The device checks every heart beat to see if the lower chambers of the heart contract in response to the delivered pulse. If no contraction is detected, a backup pace with more energy is delivered.

 April 24, 2026
April 24, 2026 









