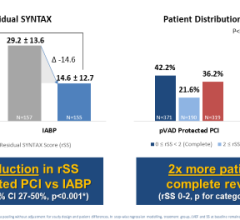
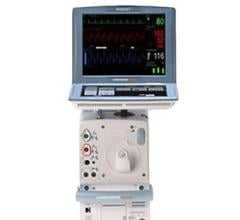
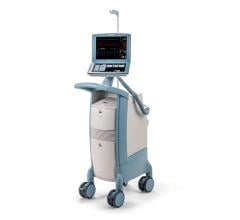
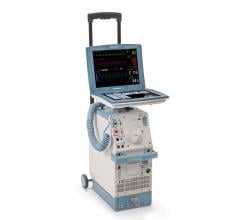
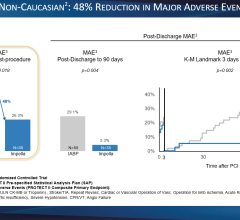
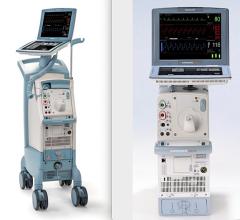
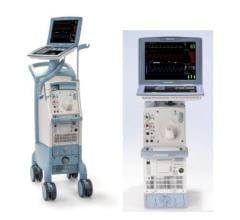
The Arrow AutoCAT 2 WAVE Interaotic balloon pump (IABP) System provides counterpulsation support to the most unstable of IABP patients, those with tachyarrhythmias and variable pulse pressures, all while maintaining 1:1 assist and with full volume delivery to the catheter.
The AutoCAT 2 WAVE IABP console, when combined with FiberOptix IAB technology, utilizes a physiologic timing algorithm called WAVE that provides uncompromised patient support to a broad range of patients.
The AutoCAT 2 WAVE IABP System with AutoPilot Operation is designed to make automatic operation adjusts in timing and triggering parameters to optimize counterpulsation support to patients under a wide range of conditions, from predictably stable to the unpredictably unstable. The company said it is the first and only IABP system that uses fiber-optic technology and a physiology-based timing algorithm, WAVE, to deliver beat-specific support.
Early inflation of the IAB within the cardiac cycle has been shown to severely compromise left ventricular function. When used with the FiberOptix IAB catheter, the AutoCAT 2 WAVE system calculates patient aortic flow on a beat-to-beat basis, instantaneously determining the aortic valve closure point for that beat. Aortic flow timing sets inflation beat to beat, automatically, matching inflation of the balloon to within 12 msec. of the time of aortic valve closure even with severe arrhythmias.
During arrhythmias, automatic R-wave deflation, coupled with aortic flow timing, delivers the closed-Loop IABP timing system. Even patients with marked arrhythmias can be more consistently supported.
For more information: www.arrowintl.com

 August 14, 2023
August 14, 2023