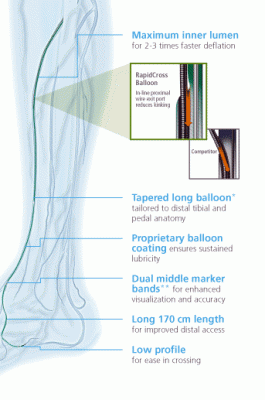
June 4, 2013 — Covidien recently introduced its RapidCross 0.014-inch Rapid Exchange Percutaneous Transluminal Angioplasty (PTA) Balloon, which offers rapid exchange (RX) convenience with kink-resistant pushability for below-the-knee (BTK) interventions.
The RapidCross balloon catheter is not a coronary device retrofitted for BTK. Every detail, from the low-profile tip to the robust RX port to the long catheter length, was thoughtfully designed for exceptional performance BTK, according to Covidien.
For more information: www.covidien.com


 June 13, 2024
June 13, 2024 









