September 3, 2014 –CorMatrix Cardiovascular announced that it has received U.S. Food and Drug Administration (FDA) clearance to market the CorMatrix CanGaroo ECM Envelope for use with cardiac implantable electronic devices (CIED’s) including pacemakers and implantable cardioverter defibrillators (ICD’s). The CorMatrix CanGaroo ECM Envelope is intended to securely hold a cardiac implantable electronic device (CIED) in order to create a stable environment when implanted in the body.
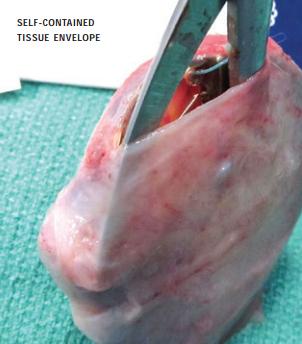
The CorMatrix CanGaroo ECM Envelope is a “pouch” which holds a CIED after implantation. It is constructed from a multi-laminate sheet of decellularized, non-crosslinked, lyophilized extracellular matrix derived from porcine small intestinal submucosa (SIS). The CorMatrix CanGaroo ECM Envelope will be provided in four sizes to fit an array of device types and sizes.
Pre-clinical testing of the CorMatrix CanGaroo ECM Envelope demonstrated that it was replaced with a vascularized tissue pouch at 90-day follow up that isolated the CIED within the normal tissue. In pre-clinical trials, the new vascularized tissue allowed for easy removal of the CIED.
“The CorMatrix CanGaroo was designed to address the needs described to us by physicians implanting CIED’s in their daily practice. We are pleased that we can provide a device that our physicians requested to address complications with CIED implantation,” said John Davis, executive vice president of sales and marketing.
For more information: www.cormatrix.com