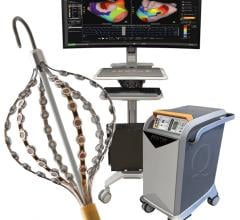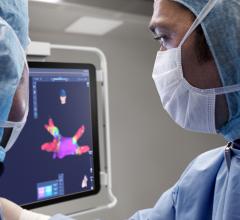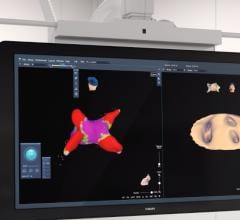
April 17, 2026 — Corify Care has announced that the U.S. Food and Drug Administration (FDA) has granted clearance for the ACORYS System, an imageless, non-invasive cardiac mapping platform capable of delivering a global, four-chamber view of cardiac electrical activity in real-time.
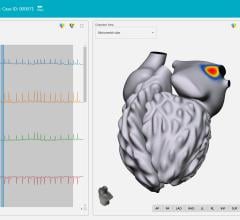
This marks a fundamental shift in electrophysiology. By combining high-resolution 3D anatomical models with real-time electrical activity, the ACORYS System introduces true 4D cardiac mapping. This instant, whole-heart functional insight enables faster, safer and more predictable ablation procedures
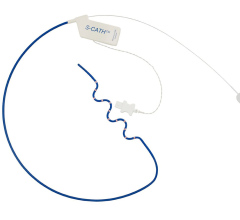
Designed to complement modern EP workflows, ACORYS empowers physicians with real-time, panoramic mapping of complex arrhythmia mechanisms. This approach allows clinical teams to gain immediate functional insight without the need for pre-procedural imaging or the induction of unstable arrhythmias.
“FDA clearance of ACORYS is a defining moment — not just for Corify, but for the future of electrophysiology,” said Andreu Climent, PhD, CEO of Corify Care. “For decades, EP has been limited by incomplete and time-intensive contact based mapping. ACORYS changes the paradigm — by delivering immediate pre-procedural and inter-procedural, global insight that allows physicians to understand complexity, define targets, and act with confidence from the very first beat.”
ACORYS provides clinicians with:
-
Single-beat, global cardiac mapping across all four chambers
-
Imageless workflow — no CT or MRI required
-
Pre-procedural, intra-procedural, and inter-procedural to assess arrhythmia complexity in real -time
-
Non-invasive functional mapping that complements standard catheter workflows
-
Rapid identification of arrhythmogenic substrate, including VT, complex atrial tachyarrhythmias or cardiac desynchronization
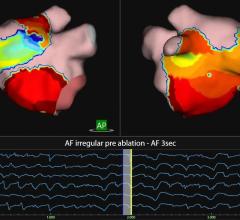
Validated in more than 2,500 patients across leading electrophysiology centers in Europe, Corify Care will present new clinical data alongside several scientific studies, at the Heart Rhythm Society (HRS) 2026 conference in Chicago. These studies along with significant prior clinical validation has demonstrated strong performance across both atrial and ventricular applications including:
-
High accuracy in identifying arrhythmia mechanism, chamber of origin, and ablation targets
-
Ability to localize ventricular tachycardia substrate in sinus rhythm, reducing the need for inducing unstable arrhythmias
-
Precise mapping of atrial activation patterns
“ACORYS gives us something we’ve never had before—a complete understanding of the arrhythmia before we even start ablating,” said Felipe Atienza, MD, CMO and Co-Founder of Corify Care. “This has the potential to dramatically change how we approach complex cases, particularly in VT and Atrial Tachyarrhythmias.”
Following FDA clearance, Corify Care will initiate a phased U.S. commercial rollout, partnering with leading electrophysiology centers to drive adoption of its next-generation mapping platform.
More information is available at www.corify.es


 September 05, 2025
September 05, 2025