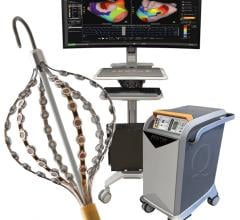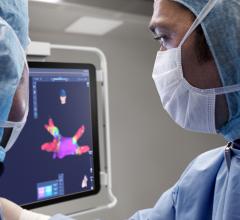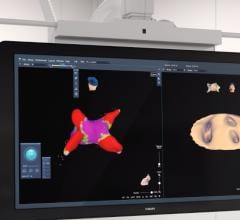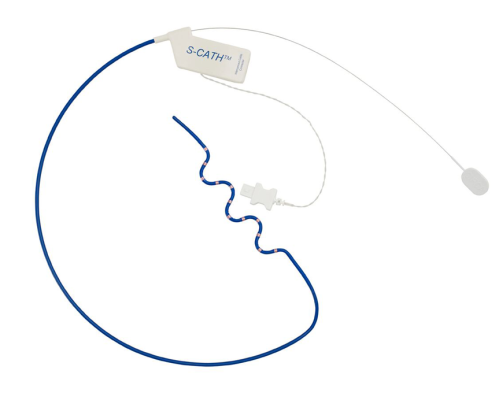
CIRCA S-CATH M esophageal temperature probe
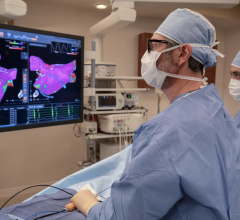
March 3, 2022 – CIRCA Scientific, Inc. has announced that its S-CATH M Esophageal Temperature Probe is now cleared for sale in the United States. The new S-CATH M Probe can be visualized on 3D Cardiac Mapping Systems, which may help physicians reduce their dependence on fluoroscopy. It is the only commercially available multi-sensor esophageal temperature probe that provides high-density, edge-to-edge coverage of the esophagus and is visible on 3D cardiac mapping systems.
Esophageal temperature probes can be used to alert physicians to critical esophageal temperature changes that may occur during cardiac ablation procedures performed in the operating room (OR) or electrophysiology (EP) lab. However, many physicians currently depend on fluoroscopy to place, position, and reposition their esophageal temperature probe during a procedure. The use of fluoroscopy requires physicians and lab personnel to wear protective lead aprons to shield themselves from radiation exposure.
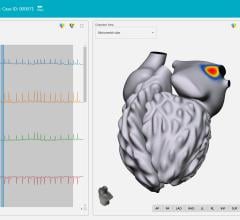
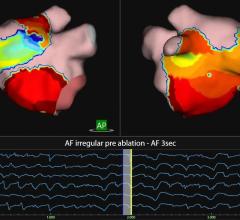
The new S-CATH M has four electrodes that are placed down the centerline of the probe, enabling it to be placed, positioned, and visualized on 3D mapping without depending solely on fluoroscopy.1 In addition, S-CATH M features a proprietary micro-electrode design that limits the amount of exposed metal to only what is absolutely necessary to be detected on 3D cardiac mapping systems, minimizing the opportunity for radiofrequency (RF) interaction. Research has shown that esophageal temperature probes with fully exposed metal electrodes may be inadvertently heated when used in close proximity to the RF field.2-4
"We designed S-CATH M to support physicians' efforts to minimize the amount of radiation their patients and staff are exposed to during their procedures without compromising on the features that set our portfolio of products apart" said Lee Geist, President, and CEO of CIRCA Scientific. "CIRCA Scientific is proud to join a growing community of medical device manufacturers meeting the demand for technology that enables reduced fluoroscopy use in the OR and EP Lab."
For more information: www.circascientific.com
References:
1 Always make sure to have a primary and secondary imaging source available, and retain the ability to verify probe location with fluoroscopy if needed.


 September 05, 2025
September 05, 2025