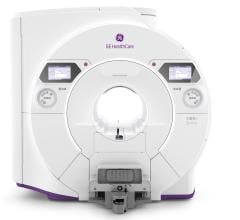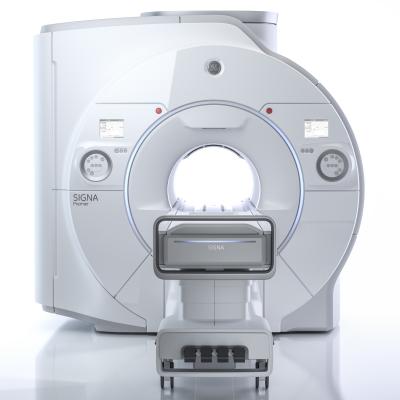
August 4, 2017 — GE Healthcare announced Signa Premier, a new wide bore 3.0T magnetic resonance imaging (MRI) system, is now available for sale in the United States. Signa Premier is the result of a four-year collaboration with the National Football League (NFL) and research institutions around the world working to design new imaging tools, particularly to aid researchers in the detection of biomarkers for the potential diagnosis of mild traumatic brain injury (TBI). GE said the system delivers a new level of clinical performance with additional research-focused capabilities, especially for neurology and oncology research.
Signa Premier features:
- GE Healthcare’s latest, short-bore, high-homogeneity 3.0T superconductive magnet;
- The most powerful gradient system GE Healthcare has ever developed for a wide bore 3.0T system, according to the company; and
- A new, digital RF transmit and receive architecture.
Clinical and research applications benefit from this high-performance hardware as well as machine learning software that includes cloud analytics. The system is powered by SuperG gradient technology. The SuperG gradient coil introduces high performance and stability, which is designed to deliver the performance of ultra-high-performance, research-class 60 centimeter MRI system in a 70 centimeter bore.
The RF technology of Signa Premier provides 146 independent receiver channels that allow the simultaneous acquisition of patient data from multiple high channel-density surface coils for faster scanning, higher image quality and overall enhanced clinical performance compared to previous-generation GE MRI systems.
The 48-channel Head Coil delivers improved clinical performance compared to previous GE MRI systems for virtually every patient. A fit-adaptable design addresses 99.99 percent of the population— including patients with extremely large heads and short necks—while preserving high signal-to-noise ratio (SNR) and high image quality. Additionally, Signa Premier can perform a routine fast brain examination in under five minutes by leveraging HyperSense, a new speed scanning tool, delivering up to eight times faster scanning than previous-generation GE MRI systems, which is part of the HyperWorks application suite.
For more information: www.gehealthcare.com


 May 14, 2026
May 14, 2026