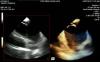
Siemens' AcuNav V 3-D intracardiac echo (ICE) catheter offers detailed, live 3-D images of the interior of the heart. This video shows an example of the catheter imaging a transseptal puncture. This new ICE technology may help better guide these punctures, which are routinely used in catheter ablations and transcatheter left atrial appendage (LAA) occluder delivery. The technology was shown as a work-in-progress during ACC 2012.
Videos
VIDEO: 3-Year Follow-up of the SYMPLICITY HTN-3 Trial
At TCT 2022, Deepak Bhatt, MD, MPh, Executive Director of Interventional Cardiovascular Programs, Brigham and Women's Hospital, presented a late-breaker session on Long-Term Outcomes Following Catheter-Based Renal Denervation in Patients With Uncontrolled Hypertensions: 3-Year Follow-up of the SYMPLICITY HTN-3 Trial. He spoke with DAIC editors about the outcomes.
Related TCT 2022 Coverage:
VIDEO: Unique Features of Coronary Artery Disease in Women
Cardiovascular Research Foundation’s (CRF) TCT 2022 in Review
Photo Gallery Highlighting TCT 2022
Cardiovascular Research Foundation’s (CRF) TCT 2022 in Review
TCT 2022 Honors Recipient of Thomas J. Linnemeier Young Investigator Award
TCT 2022 Announces Winner of Shark Tank Innovation Award
Educational Leaders Join Forces on Educational Opportunities
Top TCT 2022 Award Recipients announced by Cardiovascular Research Foundation (CRF)
EP Lab
During the American College of Cardiology (ACC) 2012 Scientific Session, McKesson displayed new features for its cardiovascular information system (CVIS). These included an electrophysiology (EP) reporting workflow, the addition of stress and Holter support and integrated inventory management. The features were shown as works-in-progress as part of the upcoming release of McKesson Cardiology v13.0, slated for release by early summer 2012. The EP reporting module integrates diagnostics including tilt tables, implantable devices and ablation treatment EP recording systems. For more information: www.allaboutCVIS.com
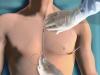
This video, provided by Boston Scientific, shows how to implant the subcutaneous implantable cardioverter defibrillator (S-ICD) system. Unlike conventional implantable cardioverter defibrillators (ICDs), which require thin, insulated wires (leads) to pass through the venous system and into the heart, the entire S-ICD System sits just below the skin and leaves the heart and blood vessels untouched. This technology has the potential to expand the reach of ICD therapy, offering physicians and appropriate patients a new alternative to traditional ICDs. Read the most recent article on the S-ICD — “FDA Clears MRI-safe Subcutaneous Implantable Cardioverter Defibrillator (S-ICD) System.”