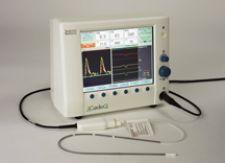
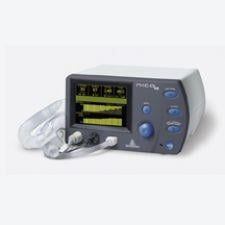
CardioQ Oesophageal Doppler Monitor from Deltex Medical
The company offers a monitor that uses a continuous wave doppler that can look right down into the aortic valve to check the size of the valve, and it can also look at the pulmonary valve. The monitor can measure stroke volume and evaluate fluid input, so as to avoid overloading the patient.
"In a matter of seconds, you can get all the information you need as often as you like with continuous monitoring," Zakar said. "You have a way to diagnose [the problem], and then you have a way to adjust the therapy and treatment."
The USCOM doppler monitor is easy to use, painless and poses a lesser risk for infection over traditional pulmonary artery catheters (PAC), Zakar said.
The USCOM doppler monitor, approved by the FDA in 2005, employs a hand-held probe whose end is the size of a dime. The probe is placed on the neck and readings can be taken and saved so physicians and nurses can recall past readings easily and quickly.
"We can teach anyone how to use this," Zakar said. "Even a parent can do it."
Cardiac output is not routinely measured in children because of technical restraints, size restrictions and reliability of data, according to USCOM. The doppler monitor is particularly useful for neonate and pediatric ICUs, said Zakar. Because children's bodies are more fragile, their systems can crash quickly, he said.
"Every day, critically ill children die because there is no [proper] measuring of hemodynamics," Zakar said.
Other approaches
British-based LiDCO takes another angle on hemodynamic monitoring. The LiDCOplus monitor focuses on the use of arterial pulse contour and power analysis to measure stroke volume. This technique measures and monitors cardiac output and stroke volume on a beat-by-beat basis from arterial pulse pressure waveforms, according to LiDCO company literature.
The advantages of this system are that it incorporates the existing arterial pressure traces in critically ill patients making its application "virtually noninvasive" and able to monitor changes in cardiac output on an almost continuous basis, according to LiDCO.
The accuracy of the pulse power technique and LiDCOplus System has been assessed in comparison to lithium dilution and PAC techniques, LiDCO states.
Another noninvasive solution is found in Respironics’ NICO2 System Respiratory Profile Monitor, which the company considers an invaluable tool for OR professionals. According to company literature, the NICO2 System allows medical staff to monitor in a noninvasive manner more than 40 variables for cardiorespiratory assessment, including cardiac output, cardiac index, stroke volume and pulmonary capillary blood flow (PCBF) to diagnose hemodynamic instability in patients.
The NICO2 Monitor measures breath-by-breath volumetric CO2 and is described by Respironics as "a revolutionary breakthrough in ventilator management ... providing faster patient feedback when it really counts."
By monitoring PCBF through capnography, the NICO2 monitor measures how effectively the heart and lungs are functioning together, giving OR physicians and nurses a more accurate measurement of cardiac output, according to company literature.
Bioreactance presented at ACC
Cheetah Medical Inc., headquartered in Israel, has developed the NICOM portable system for noninvasive, real-time cardiac output monitoring, which uses the company’s proprietary BIOREACTANCE technology designed to measure cardiac output in virtually any clinical setting.
During the convening of the American College of Cardiology in New Orleans, the company announced that an article entitled "Evaluation of a Noninvasive Continuous Cardiac Output Monitoring System Based on Thoracic Bioreactance" has been accepted for publication in the American Journal of Physiology, a leading journal in the field of cardiac physiology. (located at http://ajpheart.physiology.org/cgi/content/abstract/00195.2007v1.)
The article presents a summary of the core technology underlying its NICOM system for noninvasive cardiac output monitoring and provides validation of the ability of the system to track cardiac output in animal models under a variety of conditions and in a preliminary study in patients following cardiothoracic surgery.
Daniel Burkhoff M.D., Ph.D., article co-author and consultant to Cheetah Medical stated, "These studies provide basic validation for using the new and unique BIOREACTANCE technology used in the Cheetah NICOM system for monitoring cardiac output on a continuous basis.
"In these studies, which employed a prototype system, NICOM performed well compared to current gold standards used in the laboratory and in clinical practice. Since there is a large unmet need for cardiac output monitoring in a variety of clinical settings, these results should encourage further study and development of the clinical utility of this unique and promising technology," Dr. Burkhoff concluded.
Laura Rector, U.S. general manager for Cheetah, said, "There is a growing challenge to better address the needs of individuals suffering from congestive heart failure and hypertension. Based on the novel and unique BIOREACTANCE technology, NICOM provides a window to key cardiac function parameters, including stroke volume, thoracic fluid content and cardiac output. With BIOREACTANCE technology, common challenging factors such as electrical noise, patient movement, body habitus and electrode positioning are significantly reduced or avoided compared to other systems. "
Esophageal doppler method
The Deltex Medical CardioQ Oesophageal Doppler Monitor uses a minimally invasive approach to the routine measurement of cardiac output, allowing for the safe monitoring of fluid optimization, according to the UK-based company. The CardioQ is an esophageal doppler cardiac output and fluid management system, which the company says is "safe, easy to use and a minimally invasive alternative to the PAC."
All of the probes used with the CardioQ are sleeved in nontoxic, latex-free silicon rubber. The highly flexible nature of the probe allows it to be inserted through either the nose or mouth. The lateral stiffness, created by the design of the probe, allows the probe to be positioned easily through rotating precisely without twisting the sleeve.
"All of our probes have a single-use design; therefore, any cross-contamination risks associated with the cleaning of probes due to multiple use is removed," a Deltex Medical release states.
The CardioQ provides a determination of key cardiac function parameters in real time, based upon measurement of aortic blood flow and without the need for arterial or central venous access. Because it is a flow-based system, the CardioQ is "highly sensitive to changes" in hemodynamic status and allows for early, rapid and safe intervention, according to Deltex Medical.
In contrast to use of the PAC and techniques that rely upon pressure to determine cardiac output, the CardioQ allows the clinician to "safely, easily and reliably optimize left ventrical preload."
The CardioQ has been used on more than 300,000 patients. Deltex Medical offers case studies as examples of its clinical application.
In one case, a 31-year-old patient had been transferred to the ICU from another hospital. The initial diagnosis was sepsis secondary to pneumonia and Adult Respiratory Distress Syndrome. This patient was highly edematous and was being treated by hemofiltration therapy. The attending clinician, who had never used the CardioQ before, inserted a DP 240 Oesophageal Doppler probe orally and quickly located the descending aortic waveform. The total time for probe insertion and signal acquisition was approximately one minute. The rapid reading allowed the attending clinician to make an accurate diagnosis and make adjustments to the patient's treatment.
Accurate alternatives
According to the IHS, "What is a correct drug and/or drug combination for one patient may be totally inappropriate for another. A patient-specific therapy can be determined only from the measured, patient-specific hemodynamic state. However, since the hemodynamic state is formed for every heart beat and can be affected by a multitude of stimuli, it is important to perform a continuos hemodynamic measurement of the patient."
The noninvasive monitors offered by USCOM, LiDCO, Respironics, Cheetah and Deltex Medical provide quick, accurate readings for measuring the hemodynamic health of patients. The result is accurate diagnosis of cardiac and respiratory conditions, allowing physicians to treat conditions directly and more confidently, resulting in shorter hospital stays for patients. That translates to reduced costs for not only the patient, but healthcare facilities as well.



 April 06, 2026
April 06, 2026