The cost of bringing innovative new cardiovascular technology to the U.S. market today is staggering, numbering in the tens of millions up to $1 billion. Many in the medical device and drug industries see this as a major barrier to developing new therapies. The majority of this cost is in the preclinical animal testing stage and the human clinical trials required to gain regulatory clearance. New technologies may very soon offer a way to greatly decrease these costs. In addition, these technologies may be able to identify which drugs or device therapies will be effective in humans before spending millions in potentially dead-end research.
New technology sessions at the 2015 American College of Cardiology meeting in March highlighted some of these advances.
Heart on a Chip
The cost to develop and bring new drugs to market is astronomical, and many agents that are found to work in animal testing do not always translate into similar outcomes in human trials. This often leads to millions of dollars being spent until a first-in-human study reveals negative results, said Ashutosh Agarwal, Ph.D. assistant professor, Department of Biomedical Engineering, University of Miami, Department of Pathology, University of Miami Miller School of Medicine in Miami, Florida. He has been working on a new solution that might drastically cut costs, eliminate the need for animal studies and directly test agents in artificially grown human organ systems.
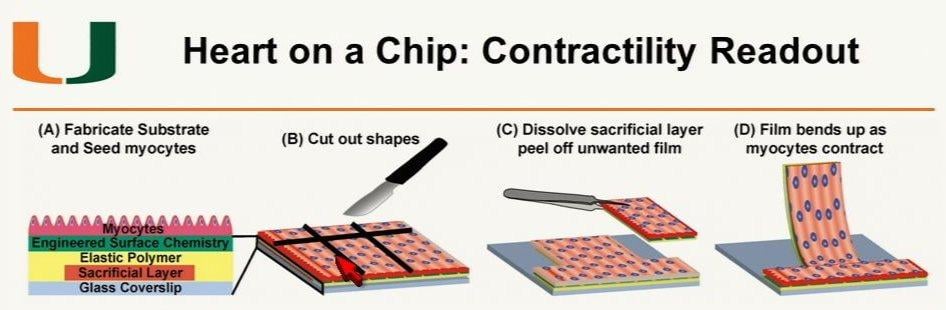
Agarwal is in charge of the Physiomimetic Microsystems Laboratory at the University of Miami, which is developing human organ mimic platforms. Often called “organs on a chip,” he explained a nano-printed silica microchip-like device can be configured in different formats to act as a scaffold for different types of human organ cell cultures. In the case of the heart, tiny tracks can be created on the chip and a culture of myocytes grow along them to create similar cellular alignment as found in the human myocardium. This “heart on a chip” can be cut into strips that will flex as the myocytes contract. Various drug candidates can be used on these chips and the contractability tested to verify if an agent has any influence on human heart tissue. Agarwal said many chips can be placed inside enclosed plates with a simulated fluid circuit system where drug agents can be introduced as if entering a patient’s blood stream. Electrophysiology tests also can be performed by wiring the chips with electrodes. He said these hearts on a chip may offer data within minutes, instead of days, weeks or months.
Currently, organs on a chip can be created to test the impact of agents on specific organs. However, Agarwal said in the future, more sophisticated systems will be created to include chips representing the various organs in the body in one connected testing system circuit to see how drug agents might effect the human body systemically.
Digital Clinical Trials
Computer modeling is a well-established way to conduct research in industries outside of healthcare and is only now starting to see application in medicine, said Paul David Morris, MBChB, MRCP, BMedSci, University of Sheffield, Sheffield, United Kingdom. Computing power today is sophisticated enough to create virtual humans that might be used in the future to perform virtual clinical trials. Sophisticated fluid dynamics software simulators have already been validated and used in other industries for virtual automotive and aviation design testing.
He said the technology already has gained acceptance with the U.S. Food and Drug Administration (FDA) with the approval last November of HeartFlow’s virtual computed tomography-fractional flow reserve (CT-FFR) software. It uses supercomputing fluid dynamics software to reconstruct a virtual representation of a patient’s myocardial blood flow from a CT angiography scan. It then creates 3-D models of the coronary arteries and assesses the FFR blood flow for all vessel segments. Prior to this FDA approval, the only way to measure FFR was using an invasive catheter-based method in the cath lab. The FDA cleared the software based on its close correlation to invasive catheter-based FFR measurements on the same patients.
Austria-based Vascops has developed similar software called A4clinics for detailed fluid dynamic analysis for aortic aneurysms, Morris said.
He explained these two companies have already demonstrated how software can accurately model blood flow in the human body, including accurate hemodynamic changes when virtual stents are placed on the models. Morris explained this could be translated into virtual clinical trials for new implantable cardiovascular devices. Morris predicts the use of so-called “in-silico” trials will be used in the coming years to help cut clinical trial costs and speed testing to bring devices to market faster.
Virtual Heart Simulators
Two examples of in-silico testing systems include complex, virtual heart software systems created by Dassault Systèmes and a joint project by Stony Brook University in New York.
Dassault, a 3-D design software company, announced the commercial release in May of its first heart model from its Living Heart Project. Powered by Dassault’s 3DExperience platform’s realistic simulation applications, the commercial, high-fidelity, scientifically validated 3-D simulator of a four-chamber human heart is the first product of its kind. With this model, device manufacturers, researchers, and medical professionals will be able to perform virtual tests and visualize the heart’s response in ways that are not possible with traditional physical testing.
The Living Heart model as delivered represents a baseline healthy heart, which can be used to study congenital defects or heart disease by modifying the shape and tissue properties in an easy-to-use software editor. In addition, medical devices can be inserted into the simulator to study their influence on cardiac function, validate their efficacy and predict reliability under a range of operating conditions. For example, coronary stents can be evaluated for optimal type, size and placement location to achieve the best performance.
“I had been aware of advances in simulation technology, but prior to the Living Heart Project I was unaware that it could address the types of challenges I have as a practicing cardiologist and medical educator,” said Robert Schwengel, M.D., FACC and clinical assistant professor of medicine, Alpert Medical School, Brown University. “Having spent time with their 3-D experiences, I believe a product like this could be very powerful in helping to educate my patients, students of medicine, and current medical professionals, as well as lead to improved diagnostic capabilities and the personalization of medical therapeutics.”
The “Living Heart” model includes well-defined anatomic details of the heart as well as proximal vasculature, such as the aortic arch, pulmonary artery and superior vena cava (SVC). The dynamic response of the heart model is governed by realistic electrical, structural and fluid (blood) flow physics.
In addition to the general availability of the heart simulator, the members of the Living Heart Project have collectively identified the highest priority cardiovascular applications for it and associated technological advancements that will help shape the functionality of future versions of the simulator.
Announced in 2014, the Living Heart Project leverages crowdsourcing of its 45 current members to build its models while protecting the intellectual property of each member. Members include regulatory science focused organizations such as the FDA and the Medical Device Innovation Consortium (MDIC), as well as technology providers, cardiologists, medical device manufacturers and hospitals such as St. Jude Medical and Mayo Clinic. This unique crowdsourcing approach has enabled the heart model to be independently tested and included in peer-reviewed scientific journals by project members, and helped Dassault deliver the first iteration of the project’s commercial product on an accelerated schedule.
“The Living Heart Project is proof that our technology can potentially change the course of therapies through simulation of the human body,” said Scott Berkey, CEO, Simulia, Dassault Systèmes. “We will continue to collaborate with the biomedical community and our partners to provide technology and applications that will enhance the experience for heart patients everywhere.”
Another virtual-heart platform proposed by Stony Brook University researchers to improve and accelerate medical-device development and testing received funding in May from the National Science Foundation (NSF). The project will receive $4.2 million over five years. The Cyberheart project is led by Scott Smolka, professor of computer science at Stony Brook University, and is part of the NSF’s center-scale initiative to advance the state-of-the-art in cyber-physical systems (CPS). CPS are engineered systems that are built from, and depend upon, the seamless integration of computation and physical components. Often called the “Internet of things,” CPS enable capabilities that go beyond the embedded systems of today. CPS such as wearable sensors and implantable devices are already being used to assess health, improve quality of life, provide cost-effective care, and potentially speed up disease diagnosis and prevention.
The Stony Brook-led project includes collaborators from seven leading universities and centers working together to develop far more realistic cardiac and device models than currently exist. This CyberHeart platform can be used to test and validate medical devices faster and at a far lower cost than existing methods. It also can be used to design optimal, patient-specific device therapies, thereby lowering the risk to the patient.
"Innovative virtual design methodologies for implantable cardiac medical devices will speed device development and yield safer, more effective devices and device-based therapies, than is currently possible," said Smolka. "We believe that our coordinated, multi-disciplinary approach, which balances theoretical, experimental and practical concerns, will yield transformational results in medical-device design and foundations of cyber-physical system verification.”
The group's approach combines patient-specific computational models of heart dynamics with advanced mathematical techniques for analyzing how these models interact with medical devices. The analytical techniques can be used to detect potential flaws in device behavior early on during the device-design phase, before animal and human trials begin. They also can be used in a clinical setting to optimize device settings on a patient-by-patient basis before devices are implanted.
Co-investigators on the project include Edmund Clarke (Carnegie Mellon University), Elizabeth Cherry (Rochester Institute of Technology), W. Rance Cleaveland (University of Maryland), Flavio Fenton (Georgia Tech), Rahul Mangharam (University of Pennsylvania), Arnab Ray (Fraunhofer Center for Experimental Software Engineering) and James Glimm and Radu Grosu (Stony Brook University). Richard A. Gray of the FDA is another key contributor.





 April 10, 2026
April 10, 2026 









