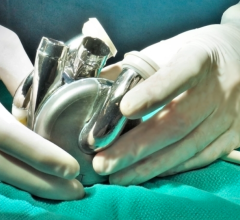
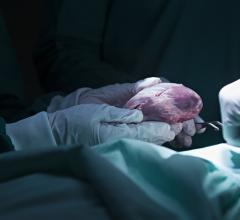
The Medtronic Heartware and Abbott Heartmate 3 LVADs and others currently cleared by the FDA might be easier to use in Medicare advanced heart failure patients under new guidelines proposed by CMS. Photo by Dave Fornell
August 21, 2020 — The U.S. Centers for Medicare and Medicaid Services (CMS) proposed updates to coverage policies for total artificial hearts (TAH) and ventricular assist devices (VAD), both of which are used to treat patients with life-threatening advanced heart failure. The agency said the changes will help expand the use of these devices to more patients and lessen the paperwork burden.
Medicare currently covers artificial hearts under the “coverage with evidence development” paradigm when beneficiaries are enrolled in a clinical study. The proposed decision memorandum would eliminate the “coverage with evidence development” requirement and provides for coverage determinations for artificial hearts to be made by local Medicare Administrative Contractors (MACs).
Although only a small number of Medicare beneficiaries receive artificial hearts, the technology can save the lives of certain end-stage heart failure patients awaiting heart transplantation. Healthcare data and analytics company GlobalData expects that the number of patients in the U.S. treated with TAHs yearly, currently standing at fewer than 300, will grow significantly if the proposed CMS coverage updates are successfully adopted. The firm said it will provide healthcare providers more flexibility and choice for treatment decisions on a patient-by-patient basis.
CMS said it believes this proposed decision is in the best interest of Medicare beneficiaries since careful patient selection is important, and the MACs are structured to take into account a beneficiary’s particular clinical circumstances to determine which patients will benefit from receiving an artificial heart.
“With about 6.5 million American adults living with heart failure, this proposal ensures a patient-centered approach to treating end-stage heart disease without delaying care,” said CMS Administrator Seema Verma. “Our updated criteria better reflects the individualized needs of patients with heart failure and expands physicians’ ability to offer the most appropriate treatment options to their patients, both of which will lead to better health outcomes for Medicare beneficiaries.”
To read the proposed decision, visit the CMS website at: https://www.cms.gov/medicare-coverage-database/details/nca-proposed-decision-memo.aspx?NCAId=298.
CMS Wants to Expand Available of VADs fro Medicare Patients
The proposed national coverage determination also provides updated coverage criteria for VADs that CMS said better aligns with current medical practice. The proposal also provides additional flexibility for patients and providers to choose the most appropriate treatments.
VADs often provide a bridge therapy to receiving a heart transplant and can be used to support the patient’s heart while it recovers on its own, or as a permanent therapy.
“VADs are a clinical treatment option for the just under 6.5 million U.S. citizens living with heart failure, as estimated by GlobalData," explained Ashley Young, medical devices analyst at GlobalData. "While heart failure currently has no cure, medications are often used to manage the disease, with VADs and TAHs being one of the last options considered. However, as VAD technology continues to improve, such as through the ability to implant the devices by lateral thoracotomy rather than open heart surgery, as was recently achieved with Medtronic’s Heartware and VAD and Abbott's HeartMate 3, VADs are continually presenting as a better and better therapeutic option over time.”
Since the proposed CMS changes will preserve patient access for all labelled VADs currently on the market, market leaders Medtronic and Abbott will be among the companies that most benefit from the coverage updates. Overall, GlobalData predicts that these coverage policy changes by the CMS will boost the VAD market in the U.S. GlobalData estimated the U.S. market value of VADs to be just under $500 million in 2019.
“VADs are often used to treat critically ill patients and procedures cannot be delayed. Many devices associated with elective procedures in the U.S. have been suffering amid the COVID-19 shutdowns as surgeries are cancelled. However, due to the nature of VAD use cases, this market likely will not be as deeply affected by the COVID-19 pandemic,” Young said.
CMS Seeking Comment on Proposed Changes on VADS and TAHs
CMS is seeking comments on the proposed national coverage determination. All public comments may be submitted at www.cms.gov/medicare-coverage-database/indexes/nca-open-for-public-comment-index.aspx. A final decision will be issued no later than 60 days after the conclusion of the 30-day public comment period.
Find more news on Heart Failure
Find more new of total artificial hearts (TAH)
Find more news on ventricular assist devices (VAD)


 April 28, 2026
April 28, 2026