March 19, 2013 — Results of the ULTIMA trial using endovascular therapy to treat submassive pulmonary embolism (PE) were presented at the American College of Cardiology 2013 meeting, The trial is the first randomized controlled trial comparing the use of an endovascular approach along with anticoagulation vs. anticoagulation therapy alone, the current standard of care.
Dr. Nils Kucher, a cardiologist at the University Hospital Bern, Switzerland, and principal investigator, reported on the 59-patient study conducted at eight and European centers. Of the 59 patients, 30 were randomly selected and treated with Ekos catheters, a device that generates ultrasound energy while simultaneously delivering a clot-dissolving drug. The advantage of this combination is that the ultrasound conditions the clot resulting in more effective drug delivery; thus ensuring faster and more complete clot dissolution. All 59 patients’ heart function were measured pre and 24 hours post therapy. Patients in the Ekos group demonstrated statistically significant reduction in right heart enlargement with no adverse effects from the catheterization. There were no serious bleeding events in either group. There was one death in the non-Ekos group for reasons other than PE.
Patients with submassive PE typically have a major clot in one or both pulmonary arteries that interferes with the heart’s ability to pump blood into the lungs for oxygenation, causing enlargement of the right side of the heart. Not removing the clot burden may quickly result in right heart failure and death, or permanent pulmonary hypertension. Anticoagulation therapy does little to dissolve clots, but rather helps prevent further clots from forming.
“ULTIMA is a pivotal study showing efficacy and safety of treating intermediate risk PE with a standardized interventional approach,” Kucher said.
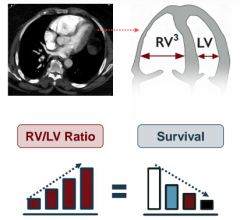
The average 24-hour improvement in right heart dysfunction among the Ekos treated patients, as measured using a heart parameter called RV/LV ratio, was 23 percent, compared to anticoagulation treated patients whose average improvement was only 3 percent. This difference is both clinically and statistically significant (p<0.0001). While patients treated with anticoagulation alone will generally improve over time, the Ekos-treated patients continued to show a statistically significant (p<0.0001) greater reduction in right heart dysfunction at 90 days than patients treated with just anticoagulation.
“It is not unlikely that the ULTIMA study may be the beginning of a treatment paradigm shift for PE similar to what was seen two decades ago for acute coronary syndrome,” Kucher added. “We used to treat acute myocardial infarction with intravenous clot dissolving drugs; yet, today this drug therapy has almost exclusively been replaced by endovascular treatment with balloons and stents. ULTIMA promises to start a similar revolution for the treatment of PE patients at increased risk of death, changing management from IV drug therapy to an endovascular approach which may be safer.”
Samuel Z. Goldhaber, M.D., professor of medicine, Harvard Medical School and director, venous thromboembolism research group, Brigham and Woman’s Hospital, Boston, and principal investigator for the SEATTLE II study. The 150-patient, single arm, prospective trial studied Ekos-treated patients suffering from massive and submassive PE. “The results of the ULTIMA trial clearly demonstrate that physicians now have another tool in their armamentarium for the safe and effective treatment of submassive PE,” he said.
For more information: www.ekoscorp.com


 March 20, 2026
March 20, 2026