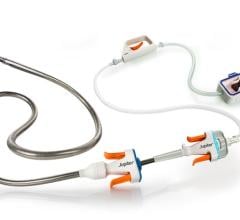
January 10, 2022 – Akura Medical Inc., a Shifamed portfolio company, announced the closing of its $25 million Series A1 financing, which will be used to support the development of its next-generation thrombectomy device. The financing was led by Cormorant Asset Management, with participation from The Capital Partnership (TCP), PA MedTech VC Fund and Shifamed angel investors.
“Venous thromboembolism (VTE) is a large market that is currently underserved due to limitations of existing treatment options,” stated Bihua Chen, Founder and Managing Member of Cormorant Asset Management. “We are excited to lead this round and believe there is ample room for improvement over existing treatments. Akura Medical’s differentiated technology has the potential to overcome these shortcomings with its elegant mechanical thrombectomy solution.”
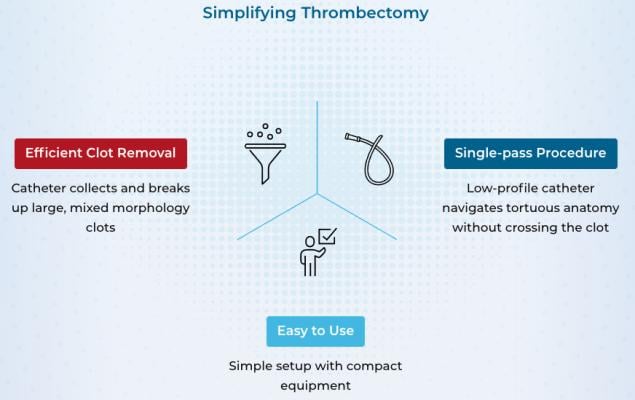
VTE includes both deep vein thrombosis (DVT), a blood clot in a deep vein, as well as pulmonary embolism (PE), which occurs when a DVT breaks free and travels to the lungs, blocking some or all blood supply. Annually in the U.S., there are roughly 900,000 VTE cases which contribute to 100,000 patient deaths. Recently, mechanical thrombectomy devices have been used to remove clots in high-risk VTE patients, however, current options are inefficient, procedurally complex, can result in significant blood loss, and have difficulty reaching the target anatomy.
“Akura Medical’s mechanical thrombectomy device is designed to simplify the procedure and integrate various mechanisms to efficiently and effectively remove blood clots,” said Amr Salahieh, founder of Shifamed and chairman of the board of Akura Medical. “We are extremely pleased to close this oversubscribed round of financing. The strong interest from investors validates the clear unmet need in this space and we look forward to expanding our world-class team as we drive towards the next development milestones.”
For more information: www.akuramed.com


 April 13, 2026
April 13, 2026