Getty Images
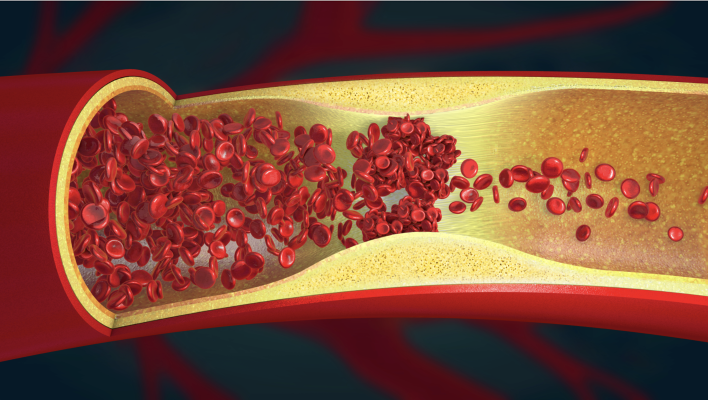
The global thrombectomy devices market is poised to experience substantial expansion, owing to the emergence of cardiovascular disorders due to the prevalence of lifestyle diseases like diabetes, unhealthy dietary habits, obesity and increasing consumption of alcohol and tobacco products. According to the WHO facts and figures, an estimated 32%, or about 17.9 million deaths worldwide, are due to cardiovascular diseases. More than 75% of these morbidities occur in low- and middle-income economies. The consistent rise in the disease as resulted in technological advancements in thrombectomy devices to meet the requirements of the expanding patient pool.
Thrombectomy devices are used for several procedures such as treating acute stroke, deep vein thrombosis (DVT), pulmonary embolism (PE) and venous thromboembolism (VTE). Numerous risks like excessive bleeding, infection, damage to the blood vessel and reaction to anesthesia associated with these procedures may hamper the industry's growth. However, the growing popularity of minimally invasive surgical interventions coupled with the increasing availability of skilled healthcare professionals and significant improvement in the healthcare infrastructure across emerging economies will create new growth avenues for the thrombectomy devices market in the years to come.
Based on a recent study by Global Market Insights Inc., the global thrombectomy devices market size may exceed $2 billion by 2028. Explained ahead are three prominent trends fueling the thrombectomy devices industry trends.
1. Growing adoption of mechanical thrombectomy.
Mechanical thrombectomy is a fast and effective remedy to treat severe medical conditions like DVT and ischemic stroke involving arterial blockages caused by blood clots.
Studies depict those patients who undergo mechanical thrombectomy have a speedy recovery and improved quality of life than those opting for other procedures like clot-busting tPAs (tissue plasminogen activators).
In April 2022, Cordis made a strategic investment in a next-gen mechanical thrombectomy device developer, E2 (Endovascular Engineering), to treat pulmonary embolism and DVT. The investment will boost the team's efforts to represent a new benchmark of care for those suffering from thromboembolism and aid the mechanical thrombectomy devices market forecast.
2. Peripheral thrombectomy devices market to gain momentum.
Peripheral or vascular thrombectomy devices will experience lucrative growth owing to the growing prevalence of peripheral arterial disorders driven by the increasing consumption of unhealthy food and smoking.
Supportive government norms and growing technological advances will further support the industry expansion during the analysis timeframe. For instance, in April 2022, Truvic Medical, an associate of Imperative Care, Inc., received a 510K approval for its Prodigy Thrombectomy System. The system is designed to treat patients with peripheral vascular thrombus and will help propel the company's development agendas toward the elevation of patient care.
3. North American region to experience robust growth.
The North America thrombectomy devices market may attract significant gains owing to the rapidly growing chronic and cardiovascular diseases patient pool and increasing geriatric population. As per the Center for Disease Control and Prevention, every one in four people, or nearly 659,000 people in the U.S., die each year from CVD.
These alarming statistics have pushed the demand for effective patient care and treatment in the region, which has increased healthcare spending substantially. This expenditure rise, along with the implementation of favorable government policies to support infrastructural development and reimbursement policies, will together boost the thrombectomy devices market share in the forthcoming years.
Related Thrombectomy Devices Content
Advances in Thrombectomy Technologies in a Post-coronary Era
Thrombectomy Market Will Grow With Newly Approved Adjustable Stent Retriever


 November 21, 2022
November 21, 2022