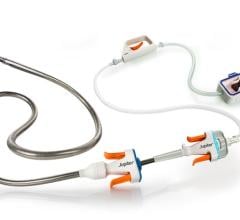
An assortment of Boston Scientific AngioJet rheolytic thrombectomy catheters. These devices use high-velocity saline jets to entrain thrombus, resulting in microfragmentation and removal.
Catheter-based blood clot removal a decade ago was a standard of care for acute coronary revascularization, but declined rapidly after several large trials showed no benefit. However today, thrombectomy is seeing increasing usage in new therapeutic areas to treat acute stoke, pulmonary embolism (PE), deep vein thrombosis (DVT) and venous thromboembolism (VTE).
Similar to cardiology in the 1980s when tissue plasminogen activator (tPA) was used as a primary method to pharmacologically revascularize patients, tPA is still the primary therapy for acute ischemic stroke. However, that trend is changing at many centers to a neurovascular interventional approach using thrombectomy catheters to remove the clot. In 2015, neurovascular thrombectomy devices made up more than half of the total market share for sales, according to Allied Market Research.[1] That share has greatly expanded since 2015 with the rapid decline of aspiration thrombectomy in percutaneous coronary interventions (PCI).
PE cases are also treated with thrombolytic therapy as the frontline standard of care, but there is increasing usage of interventional thrombectomy.
Key companies involved in making these catheters include Boston Scientific, BTG International Ltd., Medtronic, Teleflex, Penumbra Inc., Spectranetics (now part of Philips Healthcare), AngioDynamics Inc., Edwards Lifesciences, Rex Medical, Terumo, Stryker, Cerenovus and Inari Medical.
Types of Thrombectomy Devices
Catheter-directed techniques include the following:[2]
● Ultrasound-assisted thrombolysis – Catheter-directed, high-frequency ultrasound helps thrombolytic agents penetrate clots to speed the action of fibrinolytic pharmacological therapy.
● Rheolytic embolectomy – These devices inject pressurized saline through the catheter's distal tip and the macerated thrombus is aspirated through a catheter port. Examples of these devices include the AngioJet series of catheters offers by Boston Scientific.
● Rotational embolectomy – A rotating device at the catheter tip is used to fragment the clot and fragments are aspirated by the catheter.
● Aspiration thrombectomy – This includes manual clot aspiration or use of dedicated aspiration catheter devices that basically vacuum the clot out of the vessel. Examples include the Export devices from Medtronic and the QuickCat from Philips Healthcare.
● Thrombus fragmentation – Thrombus can be mechanically disrupted by manually rotating a pigtail catheter or using balloon angioplasty, but this causes small fragments of emboli to flow distally. Dedicated devices also are available, including the Cleaner from Rex Medical and the Fogarty arterial embolectomy catheter from Edwards Lifesciences.
The Abandonment of Coronary Thrombectomy
Clot aspiration was previously a widely used standard of care in patients with ST-elevation myocardial infarction (STEMI). However, recent large, randomized studies have shown very disappointing results for the routine use of thrombectomy. A meta-analysis of key trials involving 18,306 patients was published in 2017.[3] It pulled data from three large trials — TASTE, TAPAS and TOTAL.[4-6] The meta-analysis concluded routine thrombus aspiration during STEMI PCI did not improve clinical outcomes. Researchers also found aspiration may cause harm from increased stroke or transient ischemic attack (TIA) risk.
However, in patients that had a high thrombus burden, there were trends toward reduced cardiovascular death and stroke, which future trials may show benefits this subgroup.
The results of TASTE, TAPAS, TOTAL and several others led to aspiration thrombectomy being listed as offering no benefit in the 2015 ACC/AHA/SCAI Focused Update on Primary Percutaneous Coronary Intervention for Patients With ST-Elevation Myocardial Infarction.[7]
A study of data published in January 2019 online in Journal of the American Medical Association looked at aspiration data in the National Cardiovascular Data Registry CathPCI Registry.[8] The analysis of 683,584 PCI cases found thrombectomy use was associated with no difference in mortality and a small increase in in-hospital stroke. The large, nationwide analysis also showed declined aspiration thrombectomy decreased by more than 50 percent since 2011, with use as of mid-2016 at less than 5 percent.
Thrombectomy to Treat Pulmonary Embolism
While thrombolytics remains the frontline treatment, more aggressive catheter-based strategies may be useful when that therapy fails, in patients contra-indicated for anticoagulation or patients who are hemodynamically unstable.[9]
Recent advances in PE-specific systems include the 2017 U.S. Food and Drug Administration (FDA) clearance of BTG's new Ekos Control Unit 4.0. The Ekos system uses an ultrasonic catheter to generate acoustic pulses to quickly dissolve pulmonary embolism (PE), deep vein thrombosis (DVT) and peripheral arterial occlusions (PAO). The new control unit allows physicians to use one control unit to treat both pulmonary arteries. A color touch screen allows physicians to easily monitor and independently manage two Ekos devices simultaneously, simplifying bilateral PE treatment.
Related Ekos thrombectomy article, Acute Intermediate-Risk Pulmonary Embolism: A High-Stakes Conundrum
Related Ekos VIDEO: How the Ekos Thrombolytic Technology Works to Dissolve Clots
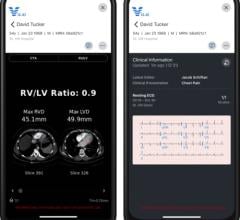
In May 2018, results from the FlowTriever Pulmonary Embolectomy (FLARE) study were presented as late-breaking clinical science at the Society for Cardiovascular Angiography and Interventions (SCAI) 2018 Scientific Sessions. The study was a prospective, single-arm multicenter trial designed to evaluate the safety and efficacy of the Inari Medical Flowtriever system in patients with acute PE. It is the first trial to treat pulmonary embolism without using thrombolytics.[10]
The study included 104 patients with acute intermediate-high risk PE treated with the device without lytics. The mean RV/LV ratio at 48 hours post-procedure decreased significantly (-0.37; standard error 0.03) from baseline (1.53; standard error 0.04). Major adverse events occurred in four patients. There were no device-related complications.
“The outcomes that we saw in this trial are extremely favorable because the improvement in the size and function of the right ventricle was comparable to previous results we’ve seen with thrombolytic therapy,” said lead author Thomas Tu, M.D., an interventional cardiologist at Baptist Health Louisville. “Our study paves the way for this novel treatment option for patients with pulmonary embolism and shows that we can decrease their risk of bleeding complications while potentially being just as safe and effective.”
Thrombectomy for Invasive Acute Treatment for Ischemic Stroke
Stroke thrombectomy makes up the majority of use for these catheter-based devices because it is a Class I indication in the 2018 Guidelines for the Early Management of Patients With Acute Ischemic Stroke from the American Heart Association/American Stroke Association.[11] The guidelines state patients should receive mechanical thrombectomy with a stent retriever if they meet all specific criteria, including being within a six-hour treatment window since the onset of symptoms.
The guideline is based on pooled patient-level data from five trials included in the Highly Effective Reperfusion Evaluated in Multiple Endovascular Stroke Trials study (HERMES), which included the MR CLEAN, ESCAPE, REVASCAT, SWIFT PRIME and EXTEND-IA trials. Among 390 patients who achieved substantial reperfusion with endovascular thrombectomy, each one-hour delay to reperfusion was associated with a less favorable degree of disability and less functional independence, but no change in mortality, the guideline states. Any cause for delay to mechanical thrombectomy, including observing for a clinical response after IV tPA, should be avoided.[11]
In January 2017, the FDA announced plans to build additional clinical evidence for stroke thrombectomy through new registries. The FDA said stroke is the fifth leading cause of death in the United States. The agency cited the same five trials used to update the 2018 stroke guidelines, suggesting a larger role for endovascular therapy. The FDA believes that research and development in this field, including the collection of data through the use of registries, provides a potential data source for expanding indications for already cleared/approved devices.
In line with the new guidelines, in February 2018, the FDA expanded the indication for Stryker's Trevo Retriever as a front-line treatment for patients experiencing acute ischemic stroke up to 24 hours from symptom onset. This increases the treatment window by 18 hours. The vendor said the expanded window has the potential to reduce disability and improve quality of life for tens of thousands of additional stroke patients each year.
"The 24-hour indication opens the treatment window to patients whose stroke would previously have progressed until all the brain tissue surrounding the affected arteries was dead, leaving them with a life of significant disabilities," said Raul Nogueira, M.D., director of neuroendovascular service and neurocritical care service, Marcus Stroke and Neuroscience Center, Grady Memorial Hospital. He also is a professor at Emory University, Atlanta. "These patients now have a much better chance for an independent life without disability."
Stryker's Trevo Retriever is the first thrombectomy device to receive FDA clearance to significantly reduce disability in patients up to 24 hours from symptom onset. Until now, mechanical thrombectomy devices like the Trevo Retriever were only cleared for use up to six hours from symptom onset. However, recent randomized clinical data from the Stryker-sponsored DAWN Trial shows that patients presenting in the six- to 24-hour window who meet specific imaging criteria and are treated with the Trevo Retriever are almost four times as likely to be functionally independent at 90 days post-stroke, compared to those treated with medical management alone.[12]
"FDA clearance of the Trevo Retriever has effectively quadrupled the treatment window from zero-six hours to zero-24 hours from time last seen well. Nonetheless, every minute still matters because in each individual patient, the earlier the blood flow is restored to the brain, the lower the resultant burden of permanent brain damage. Therefore, it is still imperative that all caregivers in the stroke pathway – from EMS to emergency room physicians to stroke neurologists to interventionalists – continue to deliver the fastest and most workflow-efficient treatment of stroke," said Tudor Jovin, M.D., co-director of the Center for Endovascular Therapy and assistant professor of neurology at the University of Pittsburgh Medical Center (UPMC).
The Trevo Retriever is a tiny stent-shaped medical device that is attached to a thin wire. In a minimally invasive procedure that utilizes X-ray, the physician navigates the retriever from the femoral artery, which is located in the upper leg, to the blocked blood artery in the brain. The retriever is designed to ensnare the blood clot and remove it from the body. Originally cleared by the FDA in 2012, the Trevo Retriever has been used in thousands of patients worldwide.
In 2018, the FDA cleared the Cerenovus Embotrap II Device, which features a dual-layer stent design that allows doctors to maintain engagement and control of a broad range of clots with minimal compression during removal. The company said about 20 percent of ischemic stroke cases remain resistant to thrombectomy due to the composition of certain blood clots.[13]
In January 2019, Cerenovus launched the EXCELLENT Registry to collect and analyze stroke-inducing blood clots removed from the brain with its Embotrap II Revascularization Device. It will enroll up to 1,000 ischemic stroke patients in as many as 50 clinical sites in the United States and Europe to gain real-world evidence. Clots also will be preserved and studied to determine different clot characteristics, including size, composition and density.
References:


 April 13, 2026
April 13, 2026