May 11, 2018 — Results of the FlowTriever Pulmonary Embolectomy (FLARE) study were presented as late-breaking clinical science at the Society for Cardiovascular Angiography and Interventions (SCAI) 2018 Scientific Sessions, April 25-28 in San Diego. The FLARE study is a prospective, single-arm multicenter trial designed to evaluate the safety and efficacy of the Flowtriever system (Inari Medical, Irvine, Calif.) in patients with acute pulmonary embolism (PE). It is the first trial to treat pulmonary embolism without using thrombolytics, the use of drugs to break up or dissolve blood clots that can lead to bleeding complications.
PE is a potentially life-threatening condition primarily caused by blood clots traveling from the veins of the legs to the lungs. It can strike anybody, young and old, healthy or sick. After coronary artery disease and stroke, acute PE ranks third among the most common types of cardiovascular diseases (NCBI). High-risk patients are often treated with catheter-directed thrombolysis (CDT), which is used to improve right ventricular function, as measured by the right ventricle to left ventricle ratio (RV/LV). However, prolonged Intensive Care Unit (ICU) stay, need for thrombolytic administration and potential bleeding complications are risks of CDT therapy. FlowTriever is the first device studied to this extent that can acutely remove a clot from the pulmonary arteries without the use of thrombolytics.
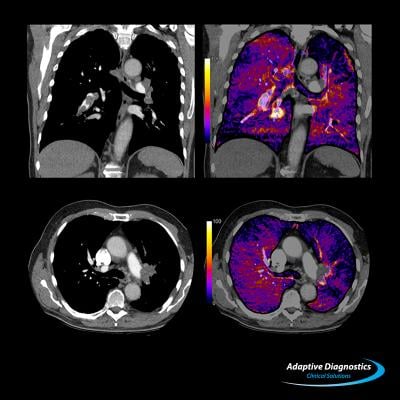
The study included 106 patients with acute intermediate-high risk PE with RV/LV ratio of 0.9 on chest computed tomography (CT). Among the 106 treated patients, two patients who received adjunctive thrombolytic therapy were analyzed separately. The primary efficacy outcome was the core-lab assessed change in RV/LV ratio measured by CT at 48 hours post-procedure. The primary safety outcome was a composite of major adverse events (MAE) — defined as device-related death, major bleeding and treatment-related adverse events (clinical deterioration, pulmonary vascular injury or cardiac injury) measured at 48 hours.
The mean RV/LV ratio at 48 hours post-procedure decreased significantly (-0.37; standard error 0.03) from baseline (1.53; standard error 0.04) (p<0.0001). MAE occurred in 4/104 (3.8 percent; 95 percent upper confidence limit 8.6 percent) patients. There were no device-related complications. One patient suffered a major bleeding complication, which was counted in the MAE. The mean procedure time was 94 ± 30 minutes. The mean ICU time post-procedure was 1.5 days, with 42 percent of patients not requiring any ICU stay.
“The outcomes that we saw in this trial are extremely favorable because the improvement in the size and function of the right ventricle was comparable to previous results we’ve seen with thrombolytic therapy,” said lead author Thomas Tu, M.D., Baptist Health Louisville. “Our study paves the way for this novel treatment option for patients with pulmonary embolism and shows that we can decrease their risk of bleeding complications while potentially being just as safe and effective.”
The authors of the study note that next steps to further assess the safety and efficacy of this treatment option is to conduct a randomized study.
Complete listing of SCAI 2018 late-breaking trials with links to articles.


 May 06, 2026
May 06, 2026 









