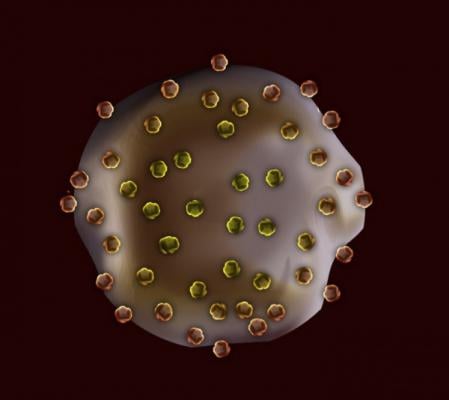
Cartoon representation of a deformable platelet-like particle with fibrin nanobodies attached
July 13, 2015 - Researchers have created tiny gel particles that can perform the same essential functions as platelets. The particles could one day be used to control excessive bleeding following traumatic injury, or in individuals with impaired clotting due to an inherited condition or as a result of certain medications or chemotherapy.
Platelets play a critical role in the body's ability to stop excess bleeding. When a blood vessel is ruptured, platelets - which normally circulate in the blood in an inactive state - become activated and aggregate at the site of the wounded vessel. During this process, a molecule called fibrinogen helps to bind the platelets together to form a temporary plug that prevents further bleeding.
While the plug is forming, a biochemical cascade is carried out that culminates in the cleavage of fibrinogen into fibrin molecules. Fibrin molecules join together to form insoluble threads, creating a natural mesh that converts the plug into a stable clot.
An important final task carried out by platelets is clot contraction. As more platelets latch on to the fibrin mesh, they begin to spread, shifting from a sphere to star-like shape. Then, over a period of several hours, platelets slowly contract.
As many platelets contract simultaneously, the clot begins to collapse, pushing out excess liquid and pulling the edges of the damaged blood vessel together. This final step is thought to be an important contributor to clot stability and wound healing.
Traditional efforts to bolster clotting have revolved around platelet transfusions, in which donor platelets are introduced into a patient's bloodstream following trauma or as a preventative measure in at-risk patients. However, once collected, donor platelets have a short shelf life (less than a week), making it hard to ensure that they will be available when patients need them. In addition, transfusions between people with incompatible blood types can occasionally result in negative side effects.
To overcome these limitations, researchers have been engineering particle-based platelet substitutes that augment clotting, are biocompatible and can be easily manufactured so as to be readily available to patients.
However, while these particle technologies have been successful in their ability to augment clotting, they have yet to overcome a key obstacle, said Thomas Barker, Ph.D., associate professor of biomedical engineering at Georgia Institute of Technology (GIT) who studies clot formation: "There's no trigger for the particle that tells it where clotting is occurring," he said.
As a result, these particles may begin to initiate clotting as soon as they enter the bloodstream, whether or not clotting is necessary and regardless of whether they're in the right spot. This kind of indiscriminate clotting can be dangerous as it can lead to blockages in vessels carrying blood to the heart, lungs, or brain.
Another limitation of current synthetic platelet substitutes is that they haven't been able to achieve some of the more complex processes carried out by platelets, such as clot contraction. This is likely due to their rigid forms, which limit their ability to change shape within the fibrin mesh clot.
With funding support from the National Institute of Biomedical Imaging and Bioengineering (NIBIB), Barker and Andrew Lyon, Ph.D., dean of the Schmid College of Science and Technology at Chapman University in California and a prior colleague of Barker's in the Chemistry Department at GIT, have created new platelet-like particles (PLPs) that home directly to the site of ongoing clotting and can induce clot contraction.
The PLPs are ultrasoft gel particles that have many fragments of antibodies - referred to as nanobodies - attached to them. These nanobodies, identified by Barker several years ago, bind specifically to fibrin, but not to fibrin's precursor, fibrinogen.
Because fibrin is only generated while a clot is being made, the PLPs only bind to and become active in areas where clotting is already ongoing; this reduces the likelihood of producing off-site clots that can be harmful.
"We had this crazy concept that you could potentially inject these particles into the bloodstream and if someone wasn't undergoing clotting, absolutely nothing would happen," said Barker.
This safeguard could make it possible for Barker and Lyon's PLPs to be given preventively to at-risk populations, such as soldiers going into battle or individuals who have impaired clotting abilities, without also increasing the risk of stroke or heart attack from unwanted clots.
Lyon, who synthesized the ultrasoft particles, says their unique ability to deform within the clot is what enables the clot to contract.
Barker and Lyon have tested their PLPs in a tiny microfluidic chamber that mimics the physiological environment inside a blood vessel, and also in an animal model of traumatic injury. They reported their results in the Sept. 7, 2014, issue of Nature Materials.
In the microfluidic chamber, PLPs that were added to platelet-deficient plasma successfully restored clotting function. In addition, PLPs restored diminished clotting function in platelet-deficient plasma samples taken from newborns who had recently undergone bypass to fix heart abnormalities.
Upon examination of PLP-induced clots, it was noted that PLPs spread significantly within the fibrin clot matrix to levels seen with natural platelets. Additional experiments also demonstrated that PLPs induce clot collapse within 24-48 hours.
While the overall degree of this collapse was similar to that found with platelet-rich plasma, it occurred at a slower rate. Finally, the researchers injected rats with PLPs five minutes prior to injury of a large blood vessel in the leg. The bleeding times of rats who had received the PLPs were significantly reduced, and this reduction was more significant than in rats who had received a 100-fold platelet infusion.
Before testing the PLPs in patients, Barker said more safety research needs to be conducted to look at whether any off-target clotting is occurring. "The technology is designed with that in mind, but designing it that way, and testing that it actually behaves how it should, are two different things," said Barker. "We need to know where the particle is going, how long it stays in the blood stream, and whether it's ever going to act poorly."
Another important question revolves around the particle's ultimate fate in the body. "The current particle is not degradable, which is what usually you want in a biomaterial," said Lyon. "We are looking at some analogs that might have better characteristics with respect to degradation. But, at the same time, the deformability of the particle may actually allow it to be distributed and excreted in a way that is very different from a rigid particle."
The team is also interested in enhancing the particles so they can secrete pro-clotting factors, an important function of native platelets. More broadly, Barker and Lyon are excited about the potential use of ultra-soft/deformable gels to develop a new class of biomaterials whose component parts more intimately interact with one another.
For more information: www.nibib.nih.gov


 October 27, 2021
October 27, 2021