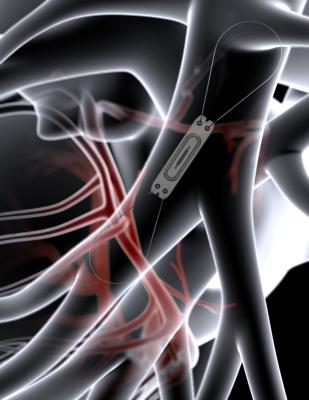
The CardioMEMS device detects worsening heart failure symptoms using pulmonary artery pressure monitoring. Pressure increases can indicate early signs of fluid backup so physicians can make changes to the patient's medications and prevent an acute congestive heart failure hospital admission.
Healthcare reform efforts of recent years have challenged the industry to reduce the costs of healthcare for both patients and providers while still maintaining a high quality of care. One of the largest drivers of cost has been repeat hospitalizations, and in 2012, the Centers for Medicare and Medicaid Services (CMS) established the Hospital Readmissions Reduction Program (HRRP) to begin solving the problem.
The HRRP seeks to reduce readmissions for specific common diseases by leveling Medicare payment reductions against hospitals that do not meet a specified target for readmission reductions. Heart failure was one of the first conditions targeted, as numerous sources say about 25 percent of patients hospitalized for heart failure are readmitted within 30 days.
Watch the VIDEO "Technologies to Reduce Heart Failure Readmissions." William Abraham, M.D., FACC, discusses advances in heart failure device treatment technologies at the Transcatheter Cardiovascular Therapeutics (TCT) 2016 annual meeting. He is director of the division of cardiovascular medicine and a professor of internal medicine, physiology and cell biology at The Ohio State University Wexner Medical Center. He also served as principal investigator of the CHAMPION Trial for the CardioMEMS device.
CardioMEMS Leads the Way
Several new technologies are being developed to remotely monitor heart failure patients for early detection of any worsening conditions to allow for early interventions and avoid hospital admissions. Arguably the highest profile of these technologies has been the CardioMEMS System from St. Jude Medical, which received U.S. Food and Drug Administration (FDA) approval in 2014. The wireless device is implanted in the pulmonary artery (PA) using a minimally invasive procedure, and then continuously measures and transmits data on PA pressure.
The CHAMPION Trial was one of the first and largest studies to assess the monitoring capabilities of the CardioMEMS device, and was the basis for FDA approval.[1] The study was a prospective, multi-center, randomized, controlled single-blind clinical trial that enrolled 550 patients at 63 U.S. sites between October 2007 and September 2009. Patients were assigned to traditional heart failure disease management, or traditional management guided by hemodynamic information from CardioMEMS. The research team used the data to adjust medication and keep patients at a PA diastolic pressure of 8-20 mmHg. Primary endpoint was heart failure hospitalizations at six months. The initial results of the CHAMPION trial demonstrated a 37 percent reduction in HF hospitalizations. The complete follow-up study of patients published in January found a 48 percent reduction in heart failure readmissions in the former control group of the trial after pulmonary artery pressure information became available to guide their therapy.[2]
“If you had a new drug that could lower heart failure readmissions by 50 percent, everyone would jump on the bandwagon and start prescribing it,” said William T. Abraham, M.D., FACP, FACC, FAHA, FESC, director, division of cardiovascular medicine, The Ohio State University, and principal investigator for the CHAMPION trial. “This technology is not as simple to use as a pill, but it is effective.”
The results were encouraging, according to Robert Bourge, M.D., Drummond Professor of Cardiovascular Medicine at the University of Alabama at Birmingham, who reviewed the trial data at the 2016 American College of Cardiology Scientific Sessions (ACC.16). Cumulative heart failure hospitalizations were consistently lower for the treatment group over the entire randomized follow-up period. A sub-analysis found that hospitalizations/readmissions also dropped among patients older than 65. Across all cases, knowledge of PA pressure derived from CardioMEMS was responsible for more than half of the documented medication changes (as opposed to changes based on signs and symptoms).
“It’s very important to realize, though, you have to be proactive with this information,” Bourge said. “It’s not a treatment. If you put the device in, you don’t look at it, you don’t act on it, you’re not going to improve your patients’ outcomes.”
Wearable Devices
In addition to implantable cardiac resynchronization therapy (CRT) devices, implantable cardioverter defibrillators (ICDs) and other implantable devices, several vendors have begun developing wearable heart failure monitors that serve many of the same functions. Several variations of this next-generation technology were highlighted at ACC.16 by Shasank Sinha, M.D., of the University of Michigan, including:
● CoVa Monitoring System by toSense. This body-worn sensor resembles a necklace and contains a pair of disposable electrodes that contact the patient’s chest. Designed to be worn for two minutes a day, the sensor measures impedance, heart rate, respiration rate and posture. Information is transmitted via Bluetooth to a gateway and then forwarded to a Web-based monitoring system. The CoVa Monitoring System received FDA approval in 2015.
● SensiVest from Sensible Medical Innovations. This wearable vest is based on military radar technology used to see through walls, but instead measures absolute lung fluid content. The first clinical study of the technology, ReDS-HF, found that the device reduced heart failure hospitalizations by 87 percent in three months compared to standard care. By tracking the remote readings, providers were able to adjust patient medication levels and return patients to the treatment goal within a week.
Watch a VIDEO showing how the SensiVest works and to hear more about it from Abraham.
● SEEQ Mobile Cardiac Telemetry System from Medtronic. This external, wireless, adhesive heart monitor transmits heart rhythm data via Bluetooth and is indicated for 30-day use. In the SEEQ study of 732 patients, the device detected a clinically relevant arrhythmia in 64 percent of patients and the average time to first clinically relevant arrhythmia was 5.8 days.
Thoracic Impedance
Studies of other heart failure remote monitoring suggest promise, but also demonstrate such technology has yet to make much of an impact on readmissions.
Several devices monitor thoracic impedance, or resistance to the flow of electricity through the heart due to fluid buildup, a possible sign of congestive heart failure (CHF). At ACC.16, Lisa Rathman, MSN, CRNP, CHFN, CCRN, lead nurse practitioner of The Heart Group of Lancaster (Pa.) General Health CHF Clinic, discussed a number of clinical trials exploring impedance monitoring devices. The MIDHeFT trial, which Rathman said was instrumental in gaining FDA approval for thoracic impedance, showed an inverse relationship between impedance and wedge pressure and that impedance does change 12-18 days ahead of a heart failure episode.
Another late-breaker presented at ACC.16, the IMPEDANCE-HF trial, found that use of the Edema Guard Monitor cut heart failure-related hospitalizations by more than half. The trial was conducted at two medical centers in Israel and included 256 patients with chronic heart failure. Heart failure hospitalizations decreased 58 percent in the treatment group during the first year, and 56 percent during the entire follow-up period. Heart failure deaths decreased 62 percent per year and all-cause deaths fell 39 percent per year.[3]
Rathman did caution that impedance can be affected by many factors and should not be taken alone as a clinical indicator of heart failure. “So I think we really have the wrong focus,” Rathman told the audience. “We’re looking at the positive predictive value of impedance. I think we should really be thinking about using impedance to risk-stratify our patients and population health management.”
Weight Monitoring
In addition to thoracic impedance, a number of technologies track weight changes for heart failure patients. A 2007 study by Sarwat Chaudhry, et al, suggested this could be a key indicator for heart failure, as it demonstrated that patients admitted for heart failure had weight increase in the days before hospitalization.[5]
Mary Walsh, M.D., FACC, medical director, heart failure and cardiac transplantation and director, nuclear cardiology at the St. Vincent Heart Center, Indianapolis, discussed this and other weight monitoring-related studies at ACC.16, including the WHARF, TEN-HMS and WISH trials. Results of all three studies indicated that weight monitoring made no significant difference in heart failure hospitalizations. The TEN-HMS Study, conducted in Europe, did indicate that overall survival was higher with telemonitoring and nurse support than with usual care alone.
Coordinated Care Programs
While technology-based monitoring efforts have had mixed results, there has been notable success with programs focused on coordinating patient follow-up. In 2010, the University of Virginia (UVA) Health System initiated a Hospital to Home (H2H) program as part of a national ACC quality initiative of the same name. The goal of H2H was to ensure that heart failure patients are seen by a physician within five to seven days after initial discharge. During the initial hospitalization, patients are thoroughly educated on medications and other aspects of home care. This care is coordinated by three nurse practitioners and ancillary staff.
To find out if the program was producing a cost savings and/or readmission reduction, UVA conducted a cost analysis study looking at four years of patient data (January 2011-December 2014). A total of 4,900 patients were admitted for heart failure during the study period; 614 (12.5 percent) of those went through the H2H program. The study examined the cost of inpatient and outpatient care at 30 days and one year post-discharge. Analysis of baseline characteristics revealed H2H patients differed from others in three key areas:
● Age (median age for H2H patients was 68 vs. 64 for all others);
● Charlson co-morbidity index (H2H patients were a little bit sicker); and
● Systolic heart failure rate (71.2 percent for H2H patients vs. 44.6 percent for non-H2H) patients.
Final analysis revealed a greater than three-fold cost savings at 30 days post-discharge ($5,767 per H2H patient vs. $21,743 per non-H2H patient) and greater than two-fold at one year ($51,343 H2H vs. $105,018 non-H2H). Mortality was also significantly reduced at both time points.
“It’s not that we’re trying to keep them out of the hospital, but we’re certainly trying to help them improve their quality of life and as a side note keep them out of the hospital,” said Timothy Welch, M.D., Fort Belvoir Community Hospital, who presented the cost analysis study in a poster presentation at ACC.16.
The state of Michigan adopted a slightly different version of the ACC Hospital-to-Home program called “See You in 7.” As with H2H, the goal of the one-year program was to have hospitalized heart failure patients see a physician within seven days of discharge. A study published in the Journal of the American College of Cardiology: Heart Failure looked at seven-day follow-up and 30-day readmission rates for the 10 hospitals participating in the “See You in 7” program and compared them to non-participants. Hospitals were evaluated on several metrics, including:
● Identifying heart failure patients prior to discharge;
● Scheduling and documenting a follow-up visit with a cardiology or primary care doctor within seven days of discharge;
● Providing patients with documentation of the scheduled follow-up;
● Identifying and addressing barriers to keeping appointment;
● Ensuring patients arrive at scheduled follow-up; and
● Making discharge summary available to follow-up healthcare providers.
Each participating hospital had a designated staff member who coordinated patient follow-up, documenting when seven-day visits occurred and investigating when they did not take place.
While seven-day follow-up rates remained low (but did improve) for both groups, participant hospitals saw 30-day readmission rates decrease a substantial 2.6 percent. Readmissions only went down 0.6 percent for non-participant hospitals.[4]
“Our study clearly shows there are challenges in coordinating early follow-up care, since increases in seven-day post-discharge follow-up were modest,” said Sandra Marie Oliver-McNeil, DNP, ACNP-BC, a study author and assistant professor of nursing at Wayne State University, Detroit. “Through collaboratively addressing the ‘See You in 7’ goals, hospitals participating in this program learned from each other when helping their patients transition from hospital to home, and they should serve as an encouraging example for other regional hospitals to share best practices.”
Watch the VIDEO "Cutting Heart Failure Length of Stay With Information Technology." Ursula Wright, MSN/MBA, FNP-BC, from Mercy Health System, explains how the nation's fifth largest Catholic health system used heart failure pathways and order sets to reduce length of stay and $14 million in costs to treat heart failure patients. Mercy earned the 2016 HIMSS Enterprise Davies Award for its leverage of information technology to impact its clinical outcomes and reduce healthcare costs.
The Debate Over 30-Day Readmissions
While much research and development has gone into reducing 30-day heart failure readmissions, many in healthcare question the validity and purpose of the metric. Those against the measure argue it does not truly signify quality healthcare and can in fact reward lower quality care. In a debate during ACC.16, Gregg Fonarow, M.D., FACC, FAHA, Eliot Corday Professor of Cardiovascular Medicine and Science at the University of California Los Angeles (UCLA), said the current readmission risk model used by Medicare is based on administrative claims data and does not adjust for numerous clinical factors. The assumption is that many readmissions are preventable, which may be the case, Fonarow said, but is not always the case. Furthermore, he argued that because current risk models do not adjust for patient characteristics, hospitals are being penalized largely based on the patients they serve rather than the quality of care provided. This includes teaching hospitals, safety-net hospitals and others serving a larger number of minority and socially disadvantaged patients.
On the pro side, Farzad Mostashari, former National Coordinator for Health Information Technology under the Obama administration, argued that readmissions are not intended to measure clinical quality. “This is a system quality measure,” he told listeners at the ACC debate. “This is a way to say the job of a hospital does not begin on admission and does not end on discharge. This is an outcome measure that encourages communication and coordination.”
While the debate over metrics continues (the majority of ACC audience members sided with Fonarow both pre- and post-debate), there is little doubt among clinicians that keeping patients out of the hospital is the end goal, and technologies and programs will likely continue to reflect this.
References:
3. Shochat, M.K., Shotan, A., Blondheim, D.S., et al. "Non-Invasive Lung IMPEDANCE-Guided Preemptive Treatment in Chronic Heart Failure Patients: A Randomized Controlled Trial (IMPEDANCE-HF Trial)," Journal of Cardiac Failure. Published online April 4, 2016. http://dx.doi.org/10.1016/j.cardfail.2016.03.015. Accessed May 27, 2016.




 April 02, 2026
April 02, 2026 









