Atrial Fibrillation
This channel includes news and new technology innovations for the treatment of atrial fibrillation, also referred to as AF or afib. AF is a cardiac arrhythmia caused by irregular and often rapid heart rate. It is caused by the upper chambers (the atria) beating irregularly and uncoordinated with the lower ventricle chambers of of the heart. Symptoms include weakness with heart palpitations and shortness of breath. The conditional can lead to an increased risk of stroke and heart failure. AF episodes can cause the blood in the atria to stagnate and form clots, usually within the left atrial appendage (LAA). The clots can flow to the brain and cause a stroke. Treatments include anticoagulation therapy to dissolve clots, catheter or surgical ablation and LAA occlusion.
When performing radiofrequency (RF) ablation to treat cardiac arrhythmia, medical professionals must balance the safety ...


For over a decade, the cardiac cryoablation industry has seen little in the way of technological advancements. Yet ...

November 8, 2013 – According to Millennium Research Group (MRG), the anticipated Food and Drug Administration (FDA) ...
September 30, 2013 — Medtronic Inc. announced clinical trial results showing that heart failure patients treated with ...
September 10, 2013 — In patients with atrial fibrillation, delayed enhancement magnetic resonance imaging (DE-MRI) ...
In the United States, the options currently available for cardiac ablation use thermal mechanisms to ablate tissue and ...
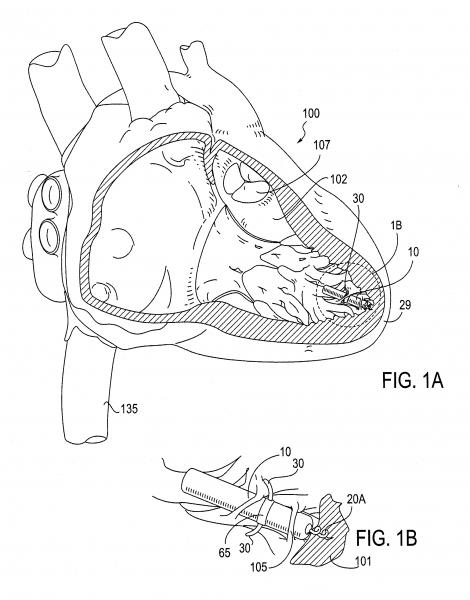
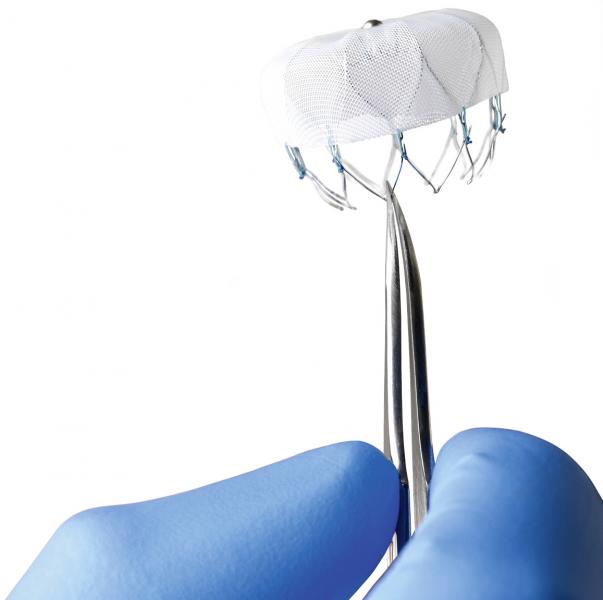
June 28, 2013 — Maquet Cardiovascular LLC announced it has acquired LAAx Inc., a privately held company that has ...
When the patients of Michael Boler, M.D. need cardiac monitoring, the Holter monitor is no longer his first choice. “The ...
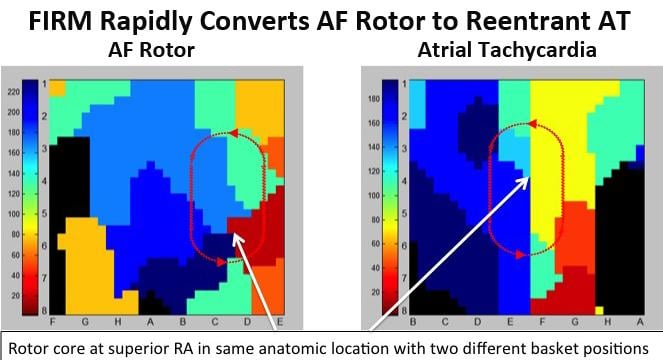
May 16, 2013 — New research presented at Heart Rhythm 2013 continues to show promising results for focal impulse and ...
May 13, 2013 – New study data released at the Heart Rhythm Society (HRS) meeting last week validates the importance of ...
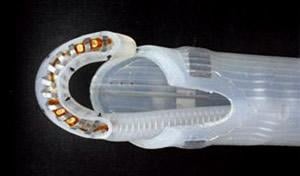
May 14, 2013 — nContact Inc. said it received conditional approval for an investigational device exemption (IDE) from ...

 December 23, 2013
December 23, 2013