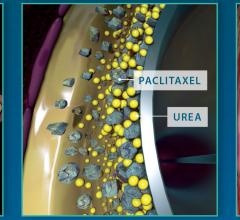
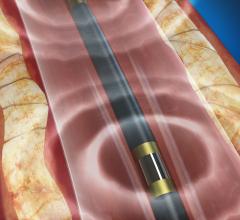
April 26, 2017 — Boston Scientific announced results from the RANGER SFA trial for the Ranger Paclitaxel-Coated PTA Ball ...
Balloon Catheter
This channel includes news and new technology innovations for angioplasty balloon catheters (PTA). These are used in arteries with atherosclerotic lesions to compress the plaque expand the artery lumen to reopen occluded or heavily stenosed atherosclerotic lesions. Balloons are often used in combination with a stent to prop the treated vessel segment open. In addition to plain old balloon angioplasty (POBA), this section includes news about drug-coated balloon (DCB), valvuloplasty balloons and specialty cutting balloon.
March 9, 2017 — Medtronic plc announced the launch of the IN.PACT BTK study to evaluate the effectiveness of a drug ...
February 8, 2017 — Medtronic plc announced receipt of an investigational device exemption (IDE) from the U.S. Food and ...
While the current positive revolution in percutaneous coronary intervention (PCI) practice has been made possible by the ...
December 6, 2016 — The Spectranetics Corp. announced in late November that its Stellarex 0.014-inch drug-coated ...
A discussion with Todd Brinton, M.D., about the newly FDA-cleared Shockwave Medical Lithoplasty System, at the ...
September 26, 2016 — C. R. Bard Inc. announced the U.S. Food and Drug Administration (FDA) approved an Investigational ...
September 22, 2016 — New data presented at the Vascular Interventional Advances (VIVA) conference demonstrated the ...
September 19, 2016 — Medtronic plc announced that the U.S. Food and Drug Administration (FDA) approved the IN.PACT ...
September 16, 2016 — The U.S. Food and Drug Administration (FDA) has granted market clearance for Shockwave Medical’s ...
This video, provided by Shockwave Medical, demonstrates the Lithoplasty System. It uses ultrasonic waves to treatment ...

Vascular Interventional Advances (VIVA) 2016 released its list late-breaking research presentations in vascular ...
July 18, 2016 — Medtronic plc has received U.S. Food and Drug Administration (FDA) approval for the IN.PACT Admiral drug ...
June 3, 2016 — Cardionovum GmbH recently announced the completion of enrollment of the RAPID trial. Results will be used ...
May 5, 2016 — Intact Vascular Inc. announced that positive twelve-month results from its Tack Optimized Balloon ...
March 28, 2016 — NuCryo Vascular LLC announced the launch of the Next Generation Cryoplasty Inflation device. The device ...

 April 26, 2017
April 26, 2017