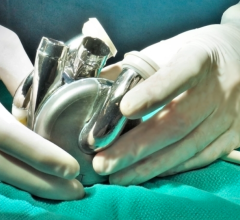
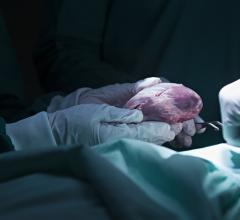
January 22, 2008 - Abiomed Inc. received FDA Humanitarian Device Exemption (HDE) supplement approval on system upgrades for its AbioCor Implantable Replacement Heart (AbioCor), intended to replace the severely damaged native heart for patients who are not eligible for a transplant and have no other treatment alternative.
The AbioCor is designed to sustain the body�s circulation and extend the lives of patients who would otherwise die of heart failure, while also offering a probable benefit for a satisfactory quality of life. The AbioCor is the first completely self-contained artificial heart that may allow patients more time at home, without wires or tubes piercing through their skin. This technology provides patients with complete mobility and remote diagnostics, according to the manufacturer.
Abiomed has selected four sites to date as AbioCor Centers including: The Johns Hopkins Hospital in Baltimore, MD; Robert Wood Johnson University Hospital in New Brunswick, NJ; Texas Heart Institute at St. Luke's Episcopal Hospital in Houston, TX; and St. Vincent's Hospital in Indianapolis, IN.
HDE approval allows the AbioCor to be made available to a limited patient population, with no more than 4,000 patients receiving the technology in the United States each year. Abiomed expects to charge $250,000 per AbioCor unit in a controlled roll-out at up to five U.S. centers.
For more information: www.abiomed.com


 April 28, 2026
April 28, 2026