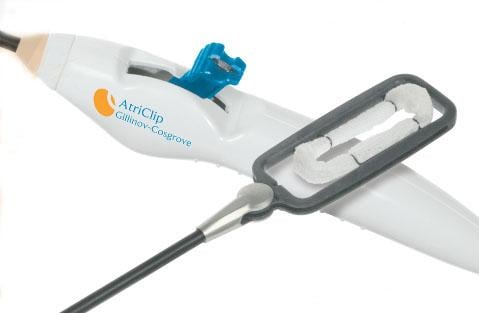
April 25, 2017 — AtriCure Inc. announced it has sold more than 100,000 AtriClip Left Atrial Appendage Exclusion System devices worldwide. This makes it the most widely used of all devices for excluding the left atrial appendage (LAA), according to the company.
The LAA is a muscular pouch attached to the heart’s left atrium. In patients with atrial fibrillation (Afib) and other cardiac arrhythmias, blood can pool and form clots in the appendage, which may then leave the heart and cause strokes. One study concluded that more than 90 percent of detected blood clots in patients with atrial fibrillation are formed in the LAA.1
Patients who suffer from Afib have a 500 percent greater risk of stroke, compared with the general population.2 Afib-related strokes are associated with higher morbidity and mortality than non-Afib related strokes.3 Prior to the invention of the AtriClip system, cardiac surgeons typically addressed the LAA during open heart surgery by cutting it off or closing off the opening of LAA to the atrium. This approach required extra time on the heart-lung machine and posed a risk of hemorrhaging and or reopening over time.
Since the launch of the AtriClip system in 2010, AtriCure has continued to expand the device’s capabilities, most recently with the AtriClip PRO2 device launched in 2016.
The AtriClip System is cleared by the U.S. Food and Drug Administration (FDA) with an indication for occlusion of the left atrial appendage, under direct visualization, in conjunction with other open cardiac surgical procedures. The clearance resulted in part from the successful EXCLUDE trial (NCT00779857), which showed the LAA was closed successfully with the AtriClip device in 98.4 percent of patients, with no device-related mortality.4
In February of 2016, AtriCure enrolled the first patient in the ATLAS (AtriClip Left Atrial Appendage Exclusion Concomitant to Structural Heart Procedures) clinical study. This observational study can enroll up to 2,000 patients without a documented pre-operative history of Afib but who present with significant risk factors for developing post-operative AF (POAF) and also have significant risk factors for bleeding on commonly prescribed medication to decrease the risk of Afib-related stroke.
This randomized prospective study (AtriClip vs. no AtriClip) will evaluate resource utilization, including hospital length of stay, emergency room and/or hospital re-admissions, and costs associated with specific adverse events that may be related to atrial fibrillation through 365 days post index procedure.
For more information: www.atricure.com
References
1. Jeff S. Healey, MD, Eugene Crystal, MD, Andrew Lamy, et al. “Left Atrial Appendage Occlusion Study (LAAOS): Results of a randomized controlled pilot study of left atrial appendage occlusion during coronary bypass surgery in patients at risk for stroke.” American Heart Journal. 2005 Aug; 150:288-93.
2. Benjamin EJ, Chen PS, Bild DE, et al. Prevention of atrial fibrillation: report from a national heart, lung, and blood institute workshop. Circulation. 2009 Feb 3; 119(4):606-18.
3. Marini C, De Santis F, Sacco S, et al. “Contribution of atrial fibrillation to incidence and outcome of ischemic stroke: results from a population-based study.” Stroke. 2005 Jun; 36 (6):1115-9.
4. Data on file at AtriCure. IDE G080095, EXCLUDE study.


 April 14, 2026
April 14, 2026 









