Getty Images
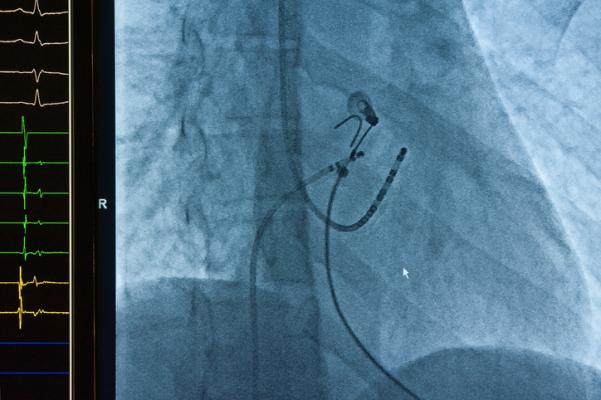
September 18, 2023 — Biosense Webster, Inc., a global leader in cardiac arrhythmia treatment and part of Johnson & Johnson MedTech,i announced today the first cases with its investigational OMNYPULSE Catheter as part of the Omny-IRE clinical trial. The first procedures were performed by Dr. Mattias Duytschaever at AZ Sint-Jan Hospital in Brugge, Belgium.ii
The Omny-IRE clinical trial is a pivotal, prospective, multi-center, non-randomized study that will enroll approximately 135 patients in Europe and Canada to evaluate the safety and effectiveness of the Biosense Webster OMNYPULSE Platform, the principal components of which are the OMNYPULSE Catheter and the TRUPULSE Generator. The study will evaluate the platform for the mapping and treatment of symptomatic paroxysmal atrial fibrillation (AFib) during standard ablation procedures.1
“Our goal at Biosense Webster is to enable electrophysiologists to deliver the best possible patient outcomes with catheter ablation; two critical components for this are integrated mapping and a versatile portfolio of tools that can address a range of workflows and patient anatomies,” said Jasmina Brooks, President, Biosense Webster. “We are working to bring forward a diverse suite of best-in-class PFA tools – including loop, focal, and large focal catheters – designed to be fully integrated with our CARTO 3 System to enable real-time catheter visualization with a low-fluoroscopy workflow.”
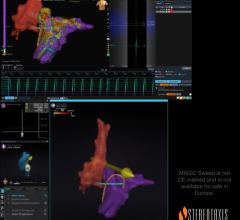
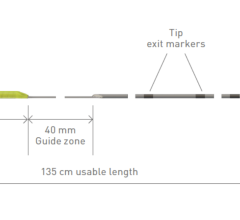
The investigational OMNYPULSE Catheter is a large-tip, 12 mm catheter with CARTO 3 System integration that is designed with contact force feedback and enhanced mapping capabilities. The investigational TRUPULSE Generator provides a bipolar, biphasic pulse sequence to the catheter’s twelve electrodes during cardiac ablation. The OMNYPULSE Platform is designed to enable AFib treatment strategies with a single device that combines a large ablation area with the ability to create high-density maps.
“In the cases I performed as part of the Omny-IRE trial, the contact force data from the OMNYPULSE Catheter aided the initial map creation in the CARTO 3 System, and the integrated PF Index module allowed me to correlate my ablation parameters with the lesion creation,” said Mattias Duytschaever M.D., Ph.D., Department of Cardiology, Electrophysiology Section, AZ Sint-Jan Hospital, Brugge, Belgium.ii “I am excited about the potential for versatility and personalization of this system, and what this means for patients.”
Catheter ablation is a minimally-invasive procedure performed by an electrophysiologist to treat heart rhythm disorders, including AFib, by interrupting irregular electrical pathways in the heart by delivering either heat (Radiofrequency ablation) or cold (Cryoablation). PFA represents a new approach to treating AFib, utilizing a controlled electric field to selectively ablate cardiac tissue that causes the irregular heart beat through a process called irreversible electroporation (IRE). Because the pulsed field energy is minimally thermal, IRE offers the potential to reduce the risk of damage to surrounding tissues including esophageal, pulmonary vein, and phrenic nerve injury.2
AFib is the most common type of cardiac arrhythmia and impacts nearly 37.5 million people worldwide,3 and 11 million people in Europe alone.4 Today, about 1 in 4 adults over the age of 40 are at risk for developing AFib.5 Despite these projections, many people are unfamiliar with AFib symptoms, available treatment options and the importance of early treatment to avoid risk of stroke and disease progression.6
“Mapping integration is critical in the treatment of AFib, especially with pulsed field energy, in order to provide immediate feedback on patient anatomy and appropriate treatment location. The ability to make optimal, patient-tailored treatment decisions with the help of an integrated solution that allows us to both evaluate and treat is of tremendous value to the EP community,” added Dr. Duytschaever.ii “I’m eager to expand our knowledge in the rapidly advancing field of PFA with the Omny-IRE trial.”
For more information: www.biosensewebster.com
References:
- Johnson & Johnson MedTech comprises the surgery, orthopedics, vision and interventional solutions businesses within Johnson & Johnson’s MedTech segment.
- AZ Sint-Jan Brugge Oostende AV entered into a clinical trial agreement with Johnson & Johnson Medical NV/SA for its participation in the Omny-IRE Clinical Trial. Dr. Mattias Duytschaever serves as a study investigator and was not compensated for his contributions to this announcement.
- ClinicalTrials.gov. A Study For Treatment of Paroxysmal Atrial Fibrillation (PAF) With the OMNYPULSE Catheter and the TRUPULSE Generator (Omny-IRE).
- Reddy VY, Neuzil P, Koruth JS, et al. Pulsed Field Ablation for Pulmonary Vein Isolation in Atrial Fibrillation. JACC 2019;74(3)315-326.
- Lippi G, Sanchis-Gomar F, Cervellin G. Global epidemiology of atrial fibrillation: An increasing epidemic and public health challenge. Int J Stroke. 2021 Feb;16(2):217-221. doi: 10.1177/1747493019897870.
- Global Burden of Disease Collaborative Network (2016) Global Burden of Disease Study 2016 (GBD 2016) Results. Seattle, United States: Institute for Health Metrics and Evaluation (IHME), 2017. Accessed 2018-04-20.
- Staerk L, et al. Lifetime risk of atrial fibrillation according to optimal, borderline, or elevated levels of risk factors: cohort study based on longitudinal data from the Framingham Heart Study. BMJ. 2018 Apr 26;361:k1453.
- Kuck et al. Catheter ablation or medical therapy to delay progression of atrial fibrillation: the randomized controlled atrial fibrillation progression trial (ATTEST). Europace 2021;23(3)362-369. PMID: 33330909.
Related Content:


 May 12, 2026
May 12, 2026