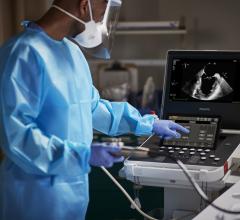
A 3-D view on the Echopixel system of a MitraClip implantation showing one clip deployed and a second about to be deployed. The images can be rotated on any axis and sliced through to show cross sectional views of the anatomy and the device in true 3-D with glasses. Echopixel is working toward a system that uses a special screen that will eliminate the need for glasses.
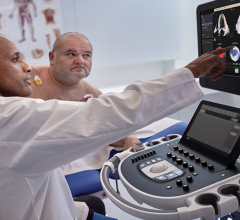
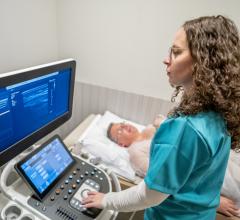
I saw a new combination of technologies on the expo floor at the American Society of Echocardiography (ASE) meeting in June that may offer a new option to potentially replace X-ray angiography (fluoroscopy) imaging systems in the cath lab. Ultrasound is already used as the primary imaging modality to guide prenatal, neonatal and some pediatric cardiac interventions. Ultrasound also is being used increasingly as a complementary imaging modality in the cath lab for soft tissue visualization and to reduce X-ray dose. In structural heart, transesophageal echo (TEE), especially 3-D/4-D TEE, has become a major aid to visualize the soft tissue landing areas for devices, navigate delivery catheters, and to evaluate securement of devices and hemodynamics prior to and immediately after deployment.
Taking the ultrasound concept a step further at ASE, GE Healthcare invited startup true 3-D visualization company Echopixel to demonstrate its technology at the GE booth. The companies actively gathered feedback from cardiologists as to its possible utility. Echopixel’s technology takes 3-D datasets from magnetic resonance imaging (MRI), computed tomography (CT) and now 3-D ultrasound and presents it in true 3-D with the use of a special display screen and 3-D glasses. The operator can use an intuitive, special stylus to latch onto the 3-D view and rotate it on any axis, or slice through the anatomy for cross-sectional views on any axis.
In recent years, there has been a lot of concern over ionizing radiation exposure from X-ray angiography imaging systems in the cath lab for both patients and clinicians. There are also inherent limitations to angiography, with the main issue being it does not show soft tissue anatomy while all the interventions it is used for navigate or implant devices in soft tissue. As transcatheter procedures become more complex, the limitations of angiography become even more apparent and are not able to meet the device guidance demands of procedures for MitraClip, septal defect occlusions, left atrial appendage (LAA) occlusions and the coming transcatheter procedures for chordae repair, annuloplasty ring repairs, and mitral and tricuspid valve implantations. Fluoroscopy has been used for interventional imaging mainly because of the lack of anything better, until now.
At ASE, GE and Echopixel showed 3-D reconstructions of LAA anatomy, mitral and aortic valves, and demonstrated a basic set of measurement tools. They also demonstrated how 3-D virtual transcatheter heart valves could be brought into the image and virtually placed to determine if the annulus/device sizing was correct and if there is the potential for paravalvular leak when the dataset was put into motion with the cardiac cycle.
The image that caught the eye of most, and made many wonder about the future of this technology, was a TEE, 3-D dynamic rendering of a MitraClip device implantation. It showed one clip already placed, a second clip attached to the mitral leaflets but still attached to the delivery catheter, and the catheter extension showing its 90 degree bend from its entry point in the atrium through a septal puncture. Wearing the 3-D glasses, it not only offers a surgical view of the implantation, but also can be rotated in any direction to see the clips and the movement of the mitral leaflets, device and catheter placement from the atrial, ventricular or transverse views. These views can also be sliced through while the heart is beating to better visualize a cross section of interest on the valve leaflets or to better see how and where the clip is engaged.
While 3-D TEE is really the technology that has enabled the possibility of performing MitraClip procedures, seeing the procedure in true, dynamic 3-D that can easily be manipulated brings in a whole new dimension. It allows the interventionalist to definitively say this is exactly where the device and leaflets are without any question. This may help greatly simplify these types of complex procedures, because it removes the guesswork and reconstruction of 3-D structures in the operator’s brain.
Echopixel and GE said they are just attempting to gauge interest in the technology and they currently cannot show or manipulate live 3-D TEE, but they said it is feasible. The glasses are also a drawback, as many interventionalists and echocardiographers do not want to wear the extra eyewear. However, Echopixel is working with a vendor that already offers true 3-D display monitors that do not require 3-D glasses. Prototypes of these screens were displayed at RSNA a couple years ago.
So, the technology components already exist to create a true 3-D/4-D interventional imaging system to better visualize soft tissue anatomy and to immediately, visually evaluate hemodynamic changes from the devices using ultrasound. Such a system could help eliminate the need for angiography in the future for transcatheter structural heart interventions. Such a system might also offer advantages of simplicity, smaller size and no ancillary concerns as with MRI, which is another proposed replacement for fluoro.
MR does not have any radiation and offers very high-quality soft tissue imaging, but it has its own set of drawbacks. The first is that the patient is inserted into the tunnel of an MRI system, limiting access to them. Second, MRI requires strict ferrous metal safety protocols, including the elimination of any items that may contain iron or steel that are not MRI safety-approved. This includes everything from wristwatches, jewelry, oxygen tanks, patient monitoring equipment, IV stands, display monitors and contrast injectors to procedural trays, and the catheters and devices used during the procedure, as well as any metal devices previously implanted anywhere inside the patient.
I see a lot of new technologies each year at the 10 or so conferences I cover, but this was one of the few that really made me stop to think about how things could be much different in clinical practice within the next decade.





 May 04, 2026
May 04, 2026