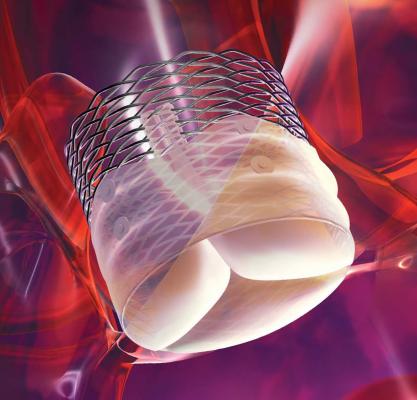
May 27, 2015 — A new study evaluating the Boston Scientific Lotus Valve System demonstrated an extremely low rate of paravalvular aortic regurgitation (leakage) for a transcatheter aortic replacement valve. Study data also showed a cardiovascular mortality rate of less than 2 percent at 30 days.
Thirty-day results for the first 250 patients in the RESPOND Post-Market Study were presented at EuroPCR 2015 in Paris by Nicolas M. Van Mieghem, M.D., co-principal investigator, Erasmus Medical Center in Rotterdam, the Netherlands. Key findings include the following:
- More than 95 percent of patients at hospital discharge had no or trace paravalvular aortic regurgitation (PVL), less than 5 percent had mild PVL and no patients exhibited moderate or severe PVL (as assessed by an independent core lab);
- The cardiovascular mortality rate was 1.6 percent at 30 days; and
- The mean pressure gradient and effective orifice area (EOA), measures used to assess the hemodynamic performance of the valve, were 10.1 +/- 3.7 mmHg and 1.9 cm2 +/- 0.4 (both p<0.001 vs. baseline).
"These first post-market study data from the RESPOND trial demonstrate that the Lotus Valve System can be used in clinical practice with an excellent safety profile and unprecedented low PVL rates," said Van Mieghem. "The absence of PVL is associated with favorable long-term survival."
In addition, rates and predictors for PVL were reported today from the REPRISE II Extended Cohort by Daniel Blackman, M.D., Leeds General Infirmary, Leeds, England. Key findings in the trial, involving 250 patients evaluated at 30 days post-implantation, include the following:
- Nearly 86 percent of patients had either no PVL or trace PVL; less than 14 percent had mild PVL and less than 1 percent had moderate PVL (as assessed by an independent core lab);
- No patients had severe PVL; and
- Significant independent predictors of PVL included device: annulus area ratio and calcium volume.
Strong performance results from both studies continue to reinforce this therapy as a less invasive treatment alternative for patients with severe aortic valve stenosis who are considered to be at high risk for surgical valve replacement.
For more information: www.bostonscientific.com


 May 06, 2026
May 06, 2026 









