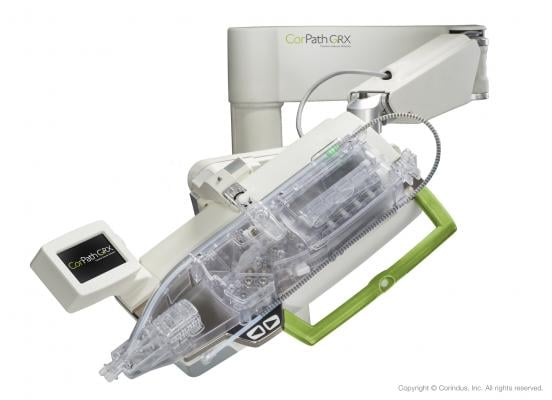
February 15, 2019 — Corindus Vascular Robotics Inc. is seeking premarket clearance from the U.S. Food and Drug Administration (FDA) to use the CorPath GRX System in neurovascular intervention. CorPath GRX, the second-generation robotic platform, received FDA clearance for percutaneous coronary intervention (PCI) in 2016 and peripheral vascular intervention (PVI) in 2018. Upon successful FDA clearance, CorPath GRX would become the world’s first and only robotic platform indicated for use in PCI, PVI and neurovascular intervention (NVI).
Corindus established a Physician Steering Committee dedicated to the Neuroendovascular sector last year and appointed Aquilla Turk, DO, as the company’s chief medical officer. Turk and the steering committee are tasked with spearheading the company’s clinical initiatives, including establishing dedicated neurovascular programs to expand robotic treatment, extending the advanced robotic capabilities across the spectrum of vascular interventions.
This announcement comes on the heels of Corindus’ announcement that its technology was successfully used to conduct the first-in-human Telerobotic Intervention Study. The study was the world’s first-in-human PCI conducted from a remote location approximately 20 miles outside of the catheterization lab.
For more information: www.corindus.com


 May 13, 2026
May 13, 2026 









