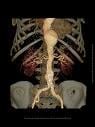
May 21, 2009 – Edwards Lifesciences Corp. last week announced the successful completion of the first human implants of its next-generation transcatheter aortic heart valve used with its new 18 Fr. delivery system.
Edwards expects these cases, performed in Canada, to be reviewed this week at EuroPCR 2009. The 18 Fr. system features the Edwards SAPIEN XT valve paired with its new, smaller NovaFlex transfemoral delivery system. The valve has a cobalt chromium alloy balloon-expandable frame, which allows for a significant reduction in its profile, and bovine pericardial tissue leaflets processed in the same stringently controlled manner as Edwards' market-leading surgical heart valves. The NovaFlex delivery system leverages the ease-of-use features of the Edwards RetroFlex line of delivery systems.
"The clinical success of the Edwards SAPIEN valve has allowed us to treat patients who previously had no good treatment options. The new Edwards SAPIEN XT valve, with its improved design and revolutionary lower profile delivery system, will allow us to treat aortic stenosis patients even more reliably and safely," said John Webb, M.D., who performed the first successful human implants of the new transcatheter system. Webb is director of the cath lab and cardiac intervention at St. Paul's Hospital in Vancouver, British Columbia, and a consultant to Edwards Lifesciences.
While the Edwards SAPIEN XT valve is being evaluated in the PREVAIL EU trial for CE mark approval, the commercial rollout of Edwards' RetroFlex 3 delivery system to be used with the Edwards SAPIEN valve is underway in Europe. The RetroFlex 3 delivery system is designed to optimize physician control of valve navigation and facilitate crossing of the patient's native calcified aortic valve.
The Edwards SAPIEN valve is approved for commercial sale in Europe. In the U.S. it is being studied as part of a randomized, pivotal clinical trial.
For more information: www.edwards.com


 May 06, 2026
May 06, 2026 









