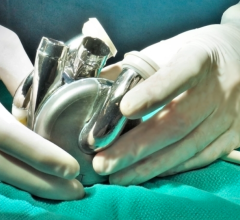
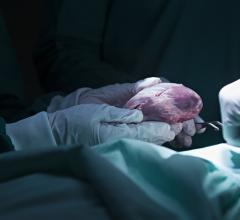
February 10, 2021 - The U.S. Food and Drug Administration (FDA) has granted artificial heart developer Carmat approval to use the new version of its artificial heart in its U.S. early feasibility study (EFS). This new version of the device includes improvements in the prosthesis and the wearable system based on clinical experience gained in the PIVOTAL study. The company believes that this latest version should further improve patient safety and quality of life.
Carmat's device is designed to fulfill an unmet medical need by providing a therapeutic alternative to heart transplants in people suffering from end-stage biventricular heart failure.
The company said it expects the first enrollments in the EFS in Q1 of 2021.
Carmat obtained the approval from the Centers for Medicare and Medicaid Services (CMS) for the reimbursement of the device and associated services within the framework of this study in May 2020.
"We are pleased to provide this new version of our artificial heart, which is similar to our CE-marked product, to the U.S. study centers," said Stéphane Piat, chief executive officer of Carmat. "Despite the COVID-19 pandemic, we were able to train and initiate three of them before the end of 2020. These centers are now ready to enroll patients. We also intend to pursue the training and initiation of additional selected centers."
The Carmat total artificial heart is composed of the implantable bioprosthesis and its portable external power supply system to which it is continuously connected. The goal for the device is to overcome the well-known shortfall in heart transplants for the tens of thousands of people suffering from irreversible end-stage heart failure, the most seriously affected of the 20 million patients with this progressive disease in Europe and the United States.
The result of combining two types of unique expertise: the medical expertise of Professor Alain Carpentier, known throughout the world for inventing Carpentier-Edwards heart valves, which are the most used in the world, and the technological expertise of Airbus Group, world aerospace leader.
The device uses highly biocompatible materials, offers a unique self-regulation system and its pulsatile nature are expected to reduce or eliminate the risk of rejection by patients bodies.
For more information: www.carmatsa.com
Related Carmat Content:
Carmat to Initiate FDA U.S. Feasibility Study of Total Artificial Heart
First Implantation of Carmat Total Artificial Heart in Denmark
Carmat Announces Start of the PIVOTAL Study


 April 28, 2026
April 28, 2026