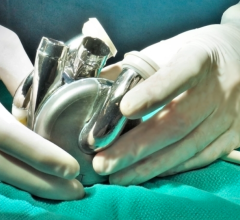
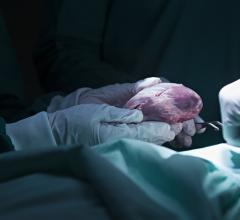
May 27, 2020 — Carmat, a developer of the of a next generation advanced total artificial heart, announces the first implantation of its bioprosthetic artificial heart in Denmark. The company aims to provide a therapeutic alternative for people suffering from end-stage biventricular heart failure.
The implant procedure was performed by the team of Dr. Peter Skov Olsen, cardiothoracic surgeon at the Heart Center of Rigshospitalet in Copenhagen. It is the first Danish hospital to implant the Carmat total artificial heart and the third international medical center contributing to the PIVOTAL study, together with the Institute for Clinical and Experimental Medicine (IKEM) in Prague, Czech Republic, and the National Research Center for Cardiac Surgery in Nur-Sultan, Kazakhstan.
Finn Gustafsson, M.D., Ph.D., professor of advanced heart failure and transplantation at the Heart Center and principal investigator of the study, comments: "We are excited to gain clinical experience with the Carmat total artificial heart (TAH) and to contribute to the PIVOTAL study. With its hemocompatibility, autoregulation and silent operation, the Cartmat TAH could become a real alternative treatment to heart transplantation for patients suffering from end-stage heart-failure."
"Rigshospitalet has developed a strong expertise in treating advanced heart failure and has participated in front-line clinical studies with innovative medical therapies and devices in this field," said Stéphane Piat, CEO of Carmat. "I am delighted that such an internationally recognized site becomes the third international center to implant our device. With the recent release of certain COVID-19 pandemic-related restrictions, Carmat's teams and proctors were able to prepare and participate in the surgical procedure and early post-operative follow up. To date, 13 patients have received our device in the PIVOTAL study and, subject to the progressive ramp-up in recruitments across our three active study centers, we estimate that 4-5 months are needed to complete enrollment and reach the total of 20 patients."
In accordance with good clinical practice and subject to regulatory obligations or specific circumstances, Carmat will not provide individual updates on patients' implantations or their health condition. However, it is planning to communicate when major milestones in the PIVOTAL study are reached, such as the opening of new centers and the overall progression of patient recruitment.
Related Carmat Artificial Heart Content:
Carmat to initiate FDA U.S. Feasibility Study of Total Artificial Heart
Carmat Announces Start of the PIVOTAL Study
VADs Gaining Popularity, But Artificial Hearts Remain the Ideal, Says GlobalData Analyst


 April 28, 2026
April 28, 2026