October 7, 2011 — Among individuals 65 years and older, as many as 30 percent have aortic valve sclerosis or stenosis and, as a result of their deteriorating health, cannot enjoy a normal lifestyle.
“The aortic valve is a gatekeeper,” says Marco Costa, M.D., of University Hospitals (UH) Case Medical in Cleveland. “If it isn’t functioning properly, there is less blood flow to the body and patients may experience fatigue, fainting, stroke, poor kidney function and heart failure. If untreated, 50 percent of individuals will die in the first year. Patients with aortic valve stenosis must be evaluated by a heart specialist to determine the best options for therapy.”
Costa is the director, Interventional Cardiovascular Center and Research & Innovation Center, UH Harrington-McLaughlin Heart & Vascular Institute at UH Case Medical Center, and professor of medicine, Case Western Reserve University School of Medicine.
Keeping Options Open
Traditional treatments for aortic valve stenosis include open surgical aortic valve replacement or palliative balloon valvuloplasty, which still may be required for some patients. The UH Institute’s heart valve center physicians, however, now have the option to replace the aortic valve using an innovative minimally invasive procedure–transcatheter aortic valve implantation (TAVI).
To support this technology, the UH Case Medical Center established the TAVI Multidisciplinary Program with a staff dedicated to providing superior care for patients who have advanced aortic valve disease. Costa leads the program along with Alan Markowitz, M.D., director, Heart Valve Center at UH Case Medical Center, and assistant professor of surgery, Case Western Reserve University School of Medicine.
Markowitz has extensive experience in surgical replacement of the aortic valve with stentless bioprostheses, and Costa has performed more than 30 TAVI procedures in Europe and eight in the United States. UH Case Medical Center recently opened state-of-the-art, hybrid operating rooms (ORs) for the cardiac catheterizations related to TAVI, and open chest surgery if necessary.
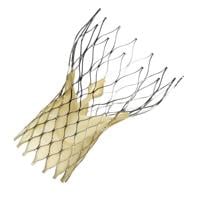
TAVI uses the expandable Medtronic CoreValve prosthesis, which is delivered through the femoral or subclavian arteries and allowed to expand into the location of the defective aortic valve.
“We are excited about our experience using the CoreValve prosthesis to treat complex aortic valve disease without removing the diseased valve,” Costa says. “This prosthesis seems superior to others because it is safe, the operator has total control, and the prosthesis can be repositioned if needed. Our program is the only one in Northeast Ohio with this capability.”
Early Success
UH Case Medical Center’s TAVI Multidisciplinary Program surgeons have among the most significant clinical experiences with implantation of the CoreValve prosthesis in the United States. As a result, they were invited to participate in Medtronic’s CoreValve U.S. Pivotal Clinical Trial. The trial is a prospective randomized study of the CoreValve prosthesis to evaluate clinical outcomes in patients with severe aortic stenosis. Surgeons at UH Case Medical Center and 40 other clinical sites around the nation will perform TAVI in approximately 1,200 patients.
“TAVI improves the quality of life for many elderly patients,” says Markowitz. “Many elderly patients may be able to participate in this study, including those 80 to 90 years of age with kidney, liver or brain diseases, and who are at risk for open heart surgery.”
“TAVI improves the quality of life for many patients,” said Costa. “The success of this procedure can lead to new treatment options for those that were almost out of possibilities.”
For more information: www.uhhospitals.org/case


 June 13, 2024
June 13, 2024 









