November 7, 2012 — Micell Technologies Inc. announced positive data from two clinical studies of its investigational MiStent Sirolimus Eluting Absorbable Polymer Coronary Stent System (MiStent SES), a thin-strut drug-eluting stent distinguished by a rapid-absorbing drug/polymer coating designed to control drug release. Eighteen-month outcomes from the DESSOLVE I trial, and nine-month data supporting all study endpoints in the DESSOLVE II trial, were presented by David Kandzari, M.D., director of interventional cardiology and chief scientific officer for the Piedmont Heart Institute of Atlanta, Ga., at the 24th annual Transcatheter Cardiovascular Therapeutics (TCT) scientific symposium.
Based on the cumulative findings and pending regulatory approval, Micell anticipates that MiStent SES may provide an innovative treatment option with an impressive safety profile for patients suffering from coronary artery disease.
"MiStent has demonstrated excellent results as well as treatment advantages we have not seen in other drug-eluting stents. Achieving minimal progression of late lumen loss from four- through 18-month assessments is unique and a significant positive clinical outcome. Once MiStent is available to physicians, this novel product will also present interventional cardiologists with a meaningful and important alternative in addressing the limitations of permanent polymer DES products. Indeed, MiStent's performance characteristics and clinical data can serve as a foundation for the improvement of patient care," said Kandzari.
Data presentations at TCT included the following:
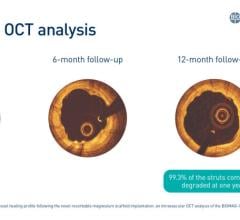
Eighteen Month Clinical and Imaging Results from the DESSOLVE I First-in-Human Trial of the MiStent SES with Absorbable Polymer: MiStent SES mean in-stent late lumen loss (LLL) was essentially unchanged through 18 months post-procedure in patients with discrete novo lesions in native coronary arteries. Initial angiographic evaluations on 25 subjects had a mean in-stent late LLL of 0.07 mm. The mean 18-month LLL for the same 25 subjects was essentially unchanged at 0.09 mm. Intravascular ultrasound (IVUS) confirmed minimal change in neointimal hyperplasia in the long-term follow-up with an initial mean (± standard deviation) percent obstruction of 7.2±4.8 percent and 11.2±8.1 percent at 18 months. The major adverse cardiac events (MACE) rate at 18 months was unchanged from 12 months, with only one non-target vessel non-Q-wave myocardial infarction (MI).
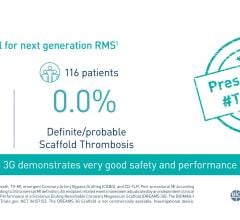
Nine Month Imaging and Twelve Month Clinical Results from the DESSOLVE II Randomized Trial of the MiStent SES with Absorbable Polymer: MiStent SES demonstrated statistically superior performance as compared to the Endeavor Sprint DES for the primary endpoint of in-stent LLL. At nine months' follow-up, in-stent LLL was 0.27 mm with a target lesion revascularization (TLR) rate of 0.9 percent. The MACE rates were 4.3 percent for MiStent SES and 6.7 percent for Endeavor.
"DESSOLVE II, a randomized controlled study, successfully met all endpoints. Patient evaluations at nine months confirmed that MiStent SES provided a healing profile comparable to that of a bare metal stent, with a median of only 0.3 percent uncovered stent struts, zero malapposition and a return of normal endothelial function with 100 percent of MiStent-treated vessels showing vasodilation. DESSOLVE II builds on the results of the first-in-human DESSOLVE I study, in which there was no increase in LLL through 18 months post-procedure," said Dennis J. Donohoe, M.D., Micell's chief medical advisor. "Each aspect of the MiStent SES was considered when designing a next-generation stent intended to deliver the efficacy of a drug-eluting stent with the safety profile of a bare metal stent. MiStent SES has the potential to address a pressing need to improve patient safety without sacrificing efficacy."
For more information: www.micell.com


 May 30, 2024
May 30, 2024