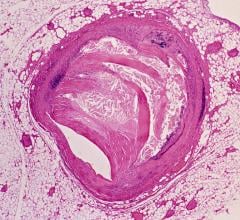
September 17, 2019 — The European Society of Cardiology (ESC) published new guidelines on the diagnosis and management of chronic coronary syndromes (CCS), which classifies computed tomography angiography (CTA) as a Class 1 recommendation for diagnosing CAD in symptomatic patients. The document, which is a continuation of the 2013 stable coronary artery disease (CAD) guidelines, updates the term to CCS to emphasize the dynamic nature of the disease. It was released at the 2019 ESC Congress, Aug. 31-Sept. 4 in Paris, France, and published simultaneously in the European Heart Journal.1
Per the guideline, unless obstructive CAD can be excluded based on clinical evaluation alone, either non-invasive functional imaging or anatomical imaging using coronary CTA should be used as the initial test to rule out or establish the diagnosis of CCS.
The new guideline suggests that "Coronary CTA is the preferred test in patients with a lower range of clinical likelihood of CAD, no previous diagnosis of CAD and characteristics associated with a high likelihood of good image quality."
“Depending on patient characteristics, local expertise and availability, as well as patient-specific considerations, physicians should decide between coronary CTA and ischemia testing,” said ESC President-elect Stephan Achenbach, M.D., FSCCT. “This is a recognition of the numerous trials that have been performed to establish the usefulness and reliability of CT angiography as a first-line examination."
“The guideline recommendations are based on data that shows that the use of coronary CTA leads to accurate identification of coronary artery disease, which if treated appropriately, can lead to improved patient outcomes.” added Ron Blankstein, M.D., FSCCT, president of SCCT. “The guidelines recognize that there are multiple factors that should be used in test selection, but that for many, a coronary CTA-first approach is beneficial.”
The guidelines also place stronger emphasis on lifestyle behaviors, and for the first time, address the negative impact of pollution and noise on CCS patients. Additionally, an annual flu vaccination, particularly for elderly patients, is recommended, as is behavioral counseling.
For more information: www.academic.oup.com/eurheartj
Reference
1. Knuuti J., Wijns W., Saraste A., et al. 2019 ESC Guidelines for the diagnosis and management of chronic coronary syndromes: The Task Force for the diagnosis and management of chronic coronary syndromes of the European Society of Cardiology (ESC). European Heart Journal, published online Aug. 31, 2019. https://doi.org/10.1093/eurheartj/ehz425

 October 24, 2025
October 24, 2025