November 7, 2022 — iRhythm Technologies, Inc., a leading cardiovascular diagnostics and digital healthcare solutions company focused on creating trusted solutions that detect, predict, and prevent disease, announced a new health economic analysis of the mSToPS study, presented at the American Heart Association’s 2022 Scientific Sessions event.
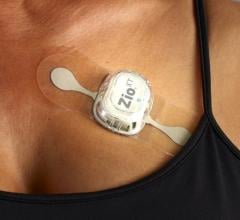
The study, titled “Cost-Effectiveness of AF Screening with Two-Week Patch Monitors: The mHealth Screening to Prevent Strokes Study,” evaluated the cost-effectiveness of screening for atrial fibrillation (AFib) with Zio XT. The analysis found that systematic screening for AFib in an at-risk population with the iRhythm Zio XT patch provided high value from a health economic perspective.
Over three years, individuals prescribed Zio XT were more likely than unmonitored individuals to have outpatient visits — including to cardiologists — but less likely to require emergency department visits or hospitalization. In patient-level projections, the group prescribed Zio XT had slightly better total and quality-adjusted survival (11.91 vs. 11.82 life years, 9.38 vs. 9.30 years). The study demonstrated an incremental cost-effectiveness ratio (ICER) of $17,000 per quality-adjusted life year gained, which is the academic standard for measuring how well medical treatments lengthen and/or improve patients' lives. The ICER is substantially lower than accepted willingness-to-pay thresholds in the U.S. and other healthcare systems. The authors concluded that screening for AFib using Zio XT provided high value from a health economic perspective.
"mSToPS is the first study to observe the benefits of AFib screening at three years — which translates to a small but meaningful gain in quality-adjusted life expectancy with a nominal (<1%) increase in lifetime healthcare spending — and the first to complete a formal health economic analysis. We conclude that AFib screening with Zio monitors is likely to both benefit older Americans and provide excellent value to the healthcare system by enabling early diagnosis," said Dr. Matthew R. Reynolds, cardiac electrophysiologist at Lahey Hospital & Medical Center and Baim Institute for Clinical Research.
mSToPS was a landmark direct-to-participant randomized clinical trial combined with a prospective matched observational cohort study. The study, published in the Journal of the American Medical Association in 2018 and conducted at the Scripps Research Translational Institute in partnership with Aetna and Janssen Pharmaceuticals, found that Zio XT-based screening was associated with increased detection of AFib, greater use of outpatient cardiology care, and a higher rate of initiation of stroke prevention therapy. A three-year follow-up analysis of the mSToPS study, published in PLOS One in 2021, found that screening with Zio XT was associated with a lower rate of clinical events and improved outcomes relative to a matched cohort.
“Taken together, these studies demonstrate screening for AFib in at-risk populations can lead to positive patient health outcomes, especially when coupled to appropriate therapy, including stroke prevention, rhythm control, and risk factor modification,” commented Dr. Mintu Turakhia, chief medical officer and chief scientific officer at iRhythm. “These studies are not in isolation and build upon a strong body of evidence from randomized trials including STROKE-STOP, EAST-AFNET 4, and others. This is precisely why we have launched Know Your Rhythm®, our risk-sharing program where we partner with healthcare systems to evaluate for undiagnosed AFib and other arrhythmias and link this to evidence-based initiation of treatment.”
For more information: https://www.irhythmtech.com/


 April 06, 2026
April 06, 2026