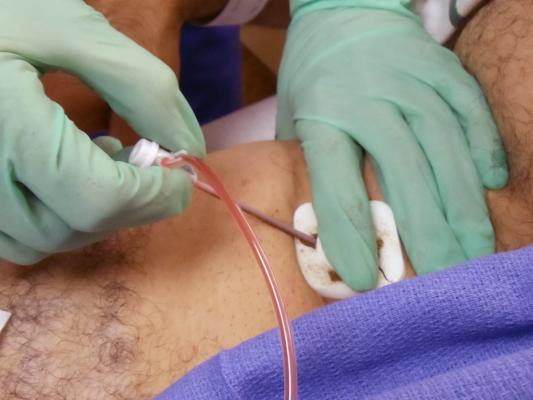
July 29, 2015 — A new clinical trial to test how a high dose of stem cells delivered via a method called retrograde coronary sinus infusion affects end-stage heart failure patients is showing promising results. The method involves giving the cells backwards (retrograde) through the heart through the main vein (the coronary sinus).
“We performed this technique because the veins do not have diseases like the arteries of the heart and it allows the cells to ‘marinate’ the entire heart at once,” explained lead investigator Amit N. Patel, M.D., MS, director of cardiovascular regenerative medicine at the University of Utah School of Medicine. The international team of researchers’ work appears in the current issue of STEM CELLS Translational Medicine.
Patients with advanced heart failure currently have few treatment options – mainly, heart transplantation or ventricular assist devices. In the search for new and effective treatments, biological-based cell and gene therapies are coming up as potential winners. They use the body's own native repair mechanisms – in particular, stem cells — to reverse or restore function in damaged organs, tissue and blood vessels.
These stem cells can come from many sources – umbilical cord, adult fat, skin tissue and more — but the most widely used source of cells when it comes to treating cardiovascular disease is the patient’s own (autologous) bone marrow. That’s because it contains the specialized stem cells that go on to become blood cells, and bone marrow is home to mesenchymal stem cells (MSCs), which are adult stem cells that can differentiate a variety of cells types depending on the conditions of the culture they are grown in.
Along with the source of stem cells used to treat heart patients, the method for delivering the cells to their proper cellular targets is another important variable affecting the success of biological-based therapy. In this study, the authors’ goal was to evaluate how retrograde bone marrow cell delivery might impact patients with either ischemic (resulting from blocked arteries, whereby the heart doesn’t receive enough blood flow and oxygen) or non-ischemic (caused by virus, high blood pressure or irregular heartbeat) heart failure.
Patel explained, “Methods such as direct injection during open heart surgery or via catheter have shown some success, but the problem is that with these methods you can only safely deliver a limited number of cells and they don’t always migrate to the areas that need repair. This makes the translation from pre-clinical models to human dosing more challenging.”
“Our previous pilot studies on patients with non end-stage heart disease indicate that retrograde delivery into the coronary sinus by infusion is successful, as it gives doctors the ability to deliver large numbers of stem cells directly to the impacted areas with minimal side effects to the patient,” he continued. The coronary sinus is accessed via the right femoral vein using a balloon infusion catheter.
However, that method had never before been tested on patients suffering from advanced heart failure. That is what Patel team’s study wanted to determine. “It’s the first clinical trial to use a large dose of nucleated bone marrow cells infused into the coronary sinus of heart failure patients,” he said.
Over the course of the 12-month trial, 60 patients were divided into two test groups according to whether they had experienced an ischemic or non-ischemic cardiac incident. Twenty-four patients in each group were given the bone marrow infusions (the BMAC groups), while the other six patients in each comprised the control group and received standard heart failure care. Patients in the BMAC groups were taken to the catheterization laboratory where both the harvest of bone marrow and infusion of cells occurred during the same visit. Patients returned for follow-up visits at one, three, six and 12 months.
“At the end of the trial, our tests revealed that all patients receiving the bone marrow infusions had improved heart function, with no adverse side effects, especially those in the non-ischemic group,” Patel reported. “As a result, we believe that this study provides the basis for a larger clinical trial in advanced heart failure patients.”
For more information: www.stemcellstm.alphamedpress.org


 May 13, 2026
May 13, 2026 









