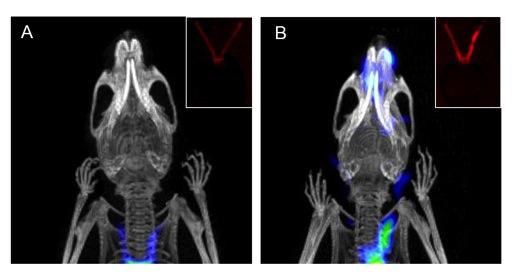
Application of dual-modality optical and PET/CT activity-based probe in experimental carotid inflammation model. Coronal noninvasive PET/CT scans of (A) healthy and (B) diseased mice with and without ligated carotid arteries respectively. Inset images show optical ex vivo florescence imagining of (A) healthy and (B) diseased carotid arteries. PET/CT and optical images courtesy of Xiaowei Ma, Toshinobu Saito and Nimali Withana.
November 1, 2016 — Researchers at Stanford University have demonstrated for the first time the use of a dual optical and positron emission tomography/computed tomography (PET/CT) activity-based probe to detect atherosclerotic plaques. The study is published in the October issue of The Journal of Nuclear Medicine.
Atherosclerosis is largely an asymptomatic disease where plaques develop over decades and symptoms do not appear until greater than 70 percent of a vessel is occluded. This results in significant risk of severe cardiovascular events such as stroke or myocardial infractions, highlighting the need for early, non-invasive diagnosis of the disease.
Matthew Bogyo, Ph.D., one of the lead authors of the study explained, “This collaborative study with Zhen Cheng, Ph.D., and Michael McConnell, M.D., provides evidence that these probes have potential benefits for non-invasive imaging of atherosclerotic plaque inflammation, potentially leading to the application of this probe in the clinic to help identify patients at high risk of developing premature atherosclerosis.”
Macrophages are cellular mediators of vascular inflammation and are involved in the formation of atherosclerotic plaques. These immune cells secrete proteases such as matrix metalloproteinases and cathepsins that contribute to disease formation and progression. In this study, activity-based probes (ABPs) targeting cysteine cathepsins were used in mouse models of atherosclerosis to non-invasively image activated macrophage populations using both optical and PET/CT methods. The probes were also used to topically label human carotid plaques, demonstrating similar specific labeling of activated macrophage populations.
The study demonstrates that ABPs targeting the cysteine cathepsins offer a rapid, non-invasive way to image atherosclerotic disease progression and plaque vulnerability.
Bogyo noted, “What’s novel about this is the fact that these probes provide accurate detection of lesions undergoing high levels of inflammatory activity and extracellular matrix remodeling. They not only enable early disease detection, they can provide real-time monitoring of therapeutic responses and clinical drug efficacy.”
He sees broader uses for the probes in the future. “The probes show efficacy in a variety of imaging modalities, including fluorescence, PET/CT, and topical application of the probe to fresh frozen murine and human tissue sections. These tools further demonstrate that the future of molecular imaging and nuclear medicine will be focused on agents that allow specific targeting of disease-associated proteins or markers that allow monitoring of disease onset, progress and response to therapeutic agents,” he said.
For more information: www.jnm.snmjournals.org


 May 12, 2026
May 12, 2026 









